Artificial enzymes perform reactions on living cells
Nature has evolved thousands of enzymes to facilitate the many chemical reactions that take place inside organisms to sustain life. Now, researchers have designed artificial enzymes that sit on the surfaces of living cells and drive reactions that could someday target drug therapies to specific organs. They report their results in the Journal of the American Chemical Society.
Metalloenzymes are a class of enzymes that contain a metal ion, such as zinc, iron or copper. The metal ion helps the enzyme speed up, or "catalyze," chemical reactions that would otherwise occur very slowly or not at all. Scientists would ultimately like to develop a method to produce therapeutic drugs only at the sites of specific cells or organs of the human body, which could reduce side effects, and enzymes could help them reach that goal. Wadih Ghattas, Jean-Pierre Mahy and their colleagues set their sights on engineering an artificial enzyme that could catalyze a useful reaction, called the Diels-Alder reaction, right on the surfaces of living cells. Chemists use this reaction to synthesize drugs, agrochemicals and many other molecules.
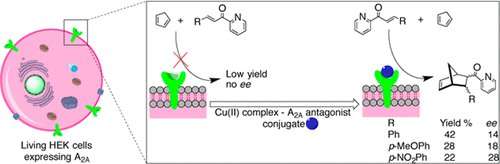
To make their artificial enzyme, the researchers began with a protein called the A2A adenosine receptor, which is naturally present on the surfaces of some cells in the body. They modified a molecule that binds to this receptor with a copper-containing chemical group that catalyzes the Diels-Alder reaction. When the researchers placed the resulting compound in a culture dish containing living human cells, it attached to the A2A adenosine receptors on the cells, forming an artificial enzyme. This enzyme catalyzed the Diels-Alder reaction with an up to 50 percent yield. The researchers say that in the future, artificial enzymes might be designed that bind to proteins found only on specific cell types, for example, cancer cells. Then, the enzyme could convert an inactive compound into a drug to selectively kill those cells.
More information: Wadih Ghattas et al. Receptor-Based Artificial Metalloenzymes on Living Human Cells, Journal of the American Chemical Society (2018). DOI: 10.1021/jacs.8b04326
Abstract
Artificial metalloenzymes are known to be promising tools for biocatalysis, but their recent compartmentalization has led to compatibly with cell components thus shedding light on possible therapeutic applications. We prepared and characterized artificial metalloenzymes based on the A2A adenosine receptor embedded in the cytoplasmic membranes of living human cells. The wild type receptor was chemically engineered into metalloenzymes by its association with strong antagonists that were covalently bound to copper(II) catalysts. The resulting cells enantioselectively catalyzed the abiotic Diels–Alder cycloaddition reaction of cyclopentadiene and azachalcone. The prospects of this strategy lie in the organ-confined in vivo preparation of receptor-based artificial metalloenzymes for the catalysis of reactions exogenous to the human metabolism. These could be used for the targeted synthesis of either drugs or deficient metabolites and for the activation of prodrugs, leading to therapeutic tools with unforeseen applications.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society