Tiny device takes big step in non-invasive fetal blood test technology

A small microfluidic device that can separate maternal and fetal cells is being developed to detect genetic abnormalities in early pregnancy through non-invasive blood tests.
The spiral-shaped plastic device is a little bigger than a matchbox and has been adapted from one initially developed to isolate tumour cells from the blood of cancer patients.
Biomedical engineers Dr. Marnie Winter and Professor Benjamin Thierry from the University of South Australia's Future Industries Institute and the ARC Centre of Excellence in Convergent Bio-Nano Science and Technology are leading a team of researchers who used the microfluidic technology to isolate fetal cells for the first time.
The special design of the microfluidic device allows large volumes of blood to be screened, paving the way for an efficient, cheap and quick method of separating fetal cells from maternal blood cells.
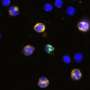
"We are separating the cells based on the size – the fetal cells are actually larger than the surrounding maternal white blood cells," Dr. Winter said.
"Then we can stain the cells for particular markers that the fetal cells express but the maternal cells don't and detect abnormalities from there."
The technology breakthrough has been published in Advanced Materials Technologies.
The device also incorporates lab-on-a-chip (LOC) technology, which integrates laboratory functions on a chip ranging in size from a few millimetres to a few square centimetres.
The UniSA researchers are working in collaboration with Dr. Majid Warkiani from the University of Technology Sydney and specialists from the Women's and Children's Hospital, SA Pathology and Repromed in South Australia.
"From about five weeks into the pregnancy, fetal cells originating from the placenta can be found in a mother's bloodstream," Dr. Winter said.
"Using modern microfluidic technology, we can now isolate these extremely rare cells (about one in a million) from the mother's white blood cells and collect them for genetic analysis.
"We are hopeful that this device could result in a new, non-invasive prenatal diagnostic test able to detect a wide range of genetic abnormalities in early pregnancy from a simple blood sample."
Non-invasive prenatal tests based on capturing fragments of fetal DNA are currently available for detecting some genetic abnormalities such as Down syndrome, Edwards syndrome (trisomy 18) and Patau syndrome (trisomy 13).
"These tests have revolutionised prenatal care, but they can only detect a small subset of genetic conditions and are not always accurate," Dr. Winter said.
"We're hopeful we would be able to detect those sorts of abnormalities too but our test would be able to give you a greater insight into the types of genetic disorders present.
"We're interested in the more complex single gene disorders such as heart conditions and other quite serious genetic abnormalities."
The research team in Adelaide is recruiting patients to get more blood samples to further develop the technology.
Dr. Winter said although a commercial product was still likely to be several years away, the technology could one day be routinely used in pregnancies as a first screening stage to flag abnormalities for further testing.
She said this would lead to more women being tested and abnormalities potentially being detected earlier in the pregnancy.
"We want to have an integrated technique where basically you could put the blood sample in and you get the result out – that's our ultimate goal," Dr. Winter said.
Professor Thierry, who leads UniSA's Bioengineering Group, said there was significant scope to further develop the lab-on-a-chip concept.
"We are collaborating with industry partners to translate this technology in routine clinical prenatal diagnostics and make it available in the future to screen low and medium-risk pregnancies," he said.
More information: Shasha Duan et al. A Highly Stretchable, Sensitive, and Transparent Strain Sensor Based on Binary Hybrid Network Consisting of Hierarchical Multiscale Metal Nanowires, Advanced Materials Technologies (2018). DOI: 10.1002/admt.201800020
Provided by The Lead