New tool analyzes disease and drug effects with unprecedented accuracy and consistency
A new protein analysis tool developed at the University at Buffalo could vastly increase the speed and precision with which disease and drug effects are analyzed.
The groundbreaking tool, called IonStar, is the first to provide near-perfect accuracy when quantifying and comparing the abundance of proteins in the bodies of people who are healthy and ill.
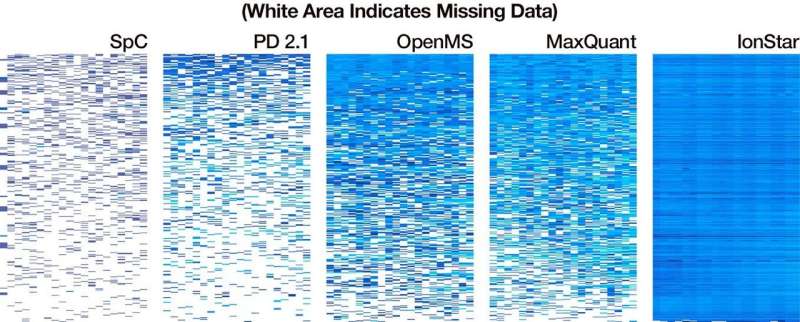
Compared to industry standard MaxQuant, IonStar improved the measurement consistency of proteins in low abundance and lowered the amount of missing data in results from 17 percent to 0.1 percent, a level that has never been achieved with large samples.
The new tool could increase the quality and accuracy of medical diagnosis and quicken the pace of pharmaceutical development.
"IonStar will totally change the face of clinical and pharmaceutical research and industry, where large investigations are often critical," says Jun Qu, Ph.D., lead investigator and professor in the UB School of Pharmacy and Pharmaceutical Sciences.
The research was published yesterday in the Proceedings of the National Academy of Sciences (PNAS).
Playing Spot the Difference
The abundance of proteins in the body that correspond with disease or pharmaceutical reactions can provide researchers with vital clues for accurately diagnosing a condition, and for developing potential therapies and evaluating drug effects.
Protein analysis tools are used to quantify and compare the abundance of proteins in groups of healthy individuals with those who are ill or treated with a drug. Changes in protein abundances, when analyzed together, often reveal novel biomarkers.
The challenge for researchers is that current tools are not efficient at analyzing large numbers of samples. One type of method, the labeling-method, uses chemical tags to label proteins. The issue: The software can only analyze up to 10 samples at a time, making it difficult for researchers to conduct typical pharmaceutical and clinical studies, says Qu.
The alternative, the label-free method, can analyze a large number of samples at once at the expense of accuracy and precision, causing researchers to waste time and resources validating falsely identified biomarkers.
IonStar increases accuracy and precision and lowers missing data by improving on sample preparation methods, alignment and feature detection designs for mass spectrometry analysis.
"For example, in clinical trials, comparing a handful of patients gets you nowhere," says Qu. "If you can analyze a large number of patients with high-quality data, you can discover and track biomarkers much more accurately and reliably. The same is true for pharmaceutical investigations."
Proving the Concept in Traumatic Brain Injury
Researchers used IonStar to quantify proteins in rats with traumatic brain injury, a debilitating condition that accounts for 2.2 million emergency room visits annually in the United States.
Using 100 tissue samples, IonStar identified 7,000 proteins, including 1,000 that differed in abundance, without missing data.
IonStar also measured low-abundance proteins with higher accuracy and precision than other prevalent analysis tools. This capability is critical, says Qu, because proteins that appear in smaller amounts play a more influential role in the body.
"If higher-level proteins are soldiers, then lower-level proteins are the commanders. They are the regulators that tell the higher-level proteins what to do," says Qu.
Qu has used IonStar and similar techniques to analyze protein variation in cancer, diabetes, cardiovascular disease, neurodegeneration and retina degeneration as well.
Future work on IonStar will focus on expanding the number of samples the tool can analyze.
More information: IonStar enables high-precision, low-missing-data proteomics quantification in large biological cohorts, Proceedings of the National Academy of Sciences (2018). DOI: 0.1073/pnas.1800541115
Journal information: Proceedings of the National Academy of Sciences
Provided by University at Buffalo