'Silent code' of nucleotides, not amino acids, determines functions of vital proteins
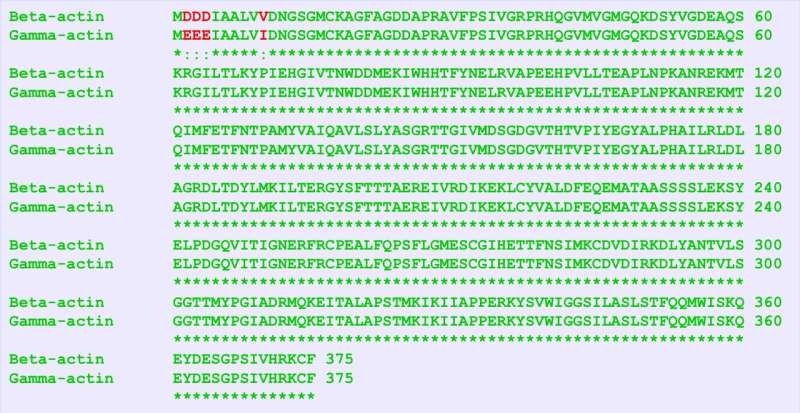
Humans possess six forms of the protein actin, which perform essential functions in the body. Two in particular, β-actin and γ-actin, are nearly identical, only differing by four amino acids. Yet these near-twin proteins carry out distinct roles. A long standing question for biologists has been, how is this possible?
"It's a mystery that's been debated in the field for the past 40 years," said Anna Kashina, a professor of biochemistry in the University of Pennsylvania School of Veterinary Medicine.
New findings by Kashina and colleagues have pointed to a surprising answer. The differing functions of these proteins are determined not by their amino acid sequences but by their genetic code.
"We like to call it the 'silent code,'" Kashina said. "Our findings show that the parts of genes that we think of as being silent actually encode very key functional information."
The researchers found that these "silent" differences in the nucleotide sequence seem to influence the density of ribosomes, the molecular machines that translate RNA into proteins. Such differences may enable each individual actin form to take on a different role in the cell.
Kashina coauthored the work, published in the journal eLife, with Penn Vet's Pavan Vedula, Satoshi Kurosaka, Nicolae Adrian Leu, Junling Wang, Stephanie Sterling and Dawei Dong and the National Institutes of Health's Yuri I. Wolf and Svetlana A. Shabalina.
Actin is so ubiquitious and essential that it's known as a "housekeeping protein." It's the most abundant protein in most cells, and its different forms play roles during cell migration, muscle contraction and development. For a time, scientists thought the different forms were functionally redundant, existing only to serve as back-ups in case one form had a defect.
More recently, researchers have come to understand that the forms are not redundant; some localize to different parts of the cells, some are incorporated into different parts of the cytoskeleton. And when these proteins are tampered with, the outcomes are different as well.
When β-actin is lacking, for example, mice die at an early stage of embryonic development. But mice lacking γ-actin, though typically smaller than normal and deaf, can survive to adulthood.
In a 2010 report in Science, Kashina's group took a step toward gaining an understanding of what determines these differences. Looking at a protein modification that normally only exists in β-actin, they found that the reason it was not also present on γ-actin was due to variations in the coding sequence between the two actin genes.
"We wanted to build on this," Kashina said, "and decided to test the hypothesis, 'What if their functional differences had nothing to do with their amino acid sequence; what if it's all in the genes?'"
The researchers took advantage of the precision gene editing made possible by the CRISPR/Cas-9 system. While the two actin isoforms differ by only four amino acids, their mRNA coding sequences differ by almost 13 percent because of "silent" nucleotide differences that nevertheless encode the same amino acids. Making changes to only five nucleotides in the β-actin gene, they were able to transform it so that its amino acid output would be exactly the same as the γ-actin protein. All that would distinguish it would be the silent nucleotide substitutions.
The gene editing worked. Mice with these edits had no β-actin protein. But unlike true β-actin knockouts they were completely healthy and viable, just as if they possessed the proper proporitions of β-actin and γ-actin proteins. They survived to reproduce and averaged the same litter sizes as normal animals.
The researchers performed the same experiment, editing the γ-actin gene to encode the β-actin protein but were only able to change the coding sequence for three of the four amino acides. Still, mice subject to this partial replacement also appeared normal and healthy, despite lacking γ-actin protein.
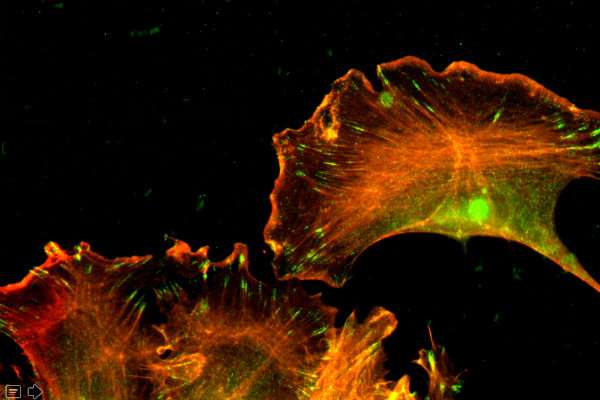
In follow-up experiments, Kashina's team found that the γ-actin proteins made from the edited β-actin gene formed a normal cytoskeleton and enabled cell to migrate in a normal fashion.
"If only the nucleotide sequence is important to protein function, then the mice shouldn't care what protein they have," Kashina said. "And the mice didn't care."
Getting at a mechanism for how DNA sequence could influence protein function, the researchers found that ribosomes density on β-actin RNA is more than a thousand times higher than on γ-actin RNA, and indeed all six actin genes had differences in ribosome density.
"This suggests that β-actin could translate into protein perhaps a thousand times faster than γ-actin," Kashina said.
And curious as to how widespread this phenomenon might be, the researchers looked for protein families with nearly identical members that are encoded by different genes and had significant variations in ribosome density across the family. They found many groups that were shared across mice, zebrafish and human genomes.
"We think this form of functional regulation is a global phenomenon," Kashina said, and it is one her lab will continue to investigate.
More information: Pavan Vedula et al, Diverse functions of homologous actin isoforms are defined by their nucleotide, rather than their amino acid sequence, eLife (2017). DOI: 10.7554/eLife.31661
F. Zhang et al. Differential Arginylation of Actin Isoforms Is Regulated by Coding Sequence-Dependent Degradation, Science (2010). DOI: 10.1126/science.1191701
Provided by University of Pennsylvania