New synthethic protocol to form 3-D porous organic network

A team of Korean researchers affiliated with UNIST has recently announced the principle of producing porous organic materials in the blink of an eye, like firing bullets. This is similar to the mechanism of chemical reaction in explosives in which pulling the trigger causes the detonator to explode.
This breakthrough has been led by Professor Jong-Beom Baek in the School of Energy and Chemical Engineering at UNIST. The study demonstrates a synthetic protocol for the formation of a three-dimensional porous organic network via solid-state explosion of organic single crystals.
Three-dimensional porous materials have high surface area, which could be useful for applications such as catalytic supports, gas capture and storage, energy conversion and storage, optoelectronics and semiconductors. Typical examples are zeolites and zeolite-like materials. However, in recent years, extensive studies have been conducted to produce porous materials from more durable organic materials. And they make use of a synthetic approach to produce a 3-D porous organic network in liquid-phase reactions in the presence of suitable solvents and/or catalysts. However, the resultant products are of low purity, and therefore, post-treatment for purification becomes necessary.
In the study, Professor Baek and his team introduced a new synthetic methodology for the fabrication of a 3-D porous organic network with high specific surface area via solid-state explosion of organic single crystals containing primer molecules. The explosive reaction is realized by the Bergman reaction (cycloaromatization) of three enediyne groups on 2,3,6,7,14,15-hexaethynyl-9,10-dihydro-9,10-[1,2]benzenoanthracene (HEA), which is a self-polymerizable trifunctional (M3) building block with three enediyne groups (containing a double bond and two triple bonds). In general, solid-organic materials easily can melt when heat is applied. However, their newly developed HEA single crystals trigger an explosive Bergman reaction and quickly change to 3-D porous materials without the presence of solvents and catalysts when heat is applied.
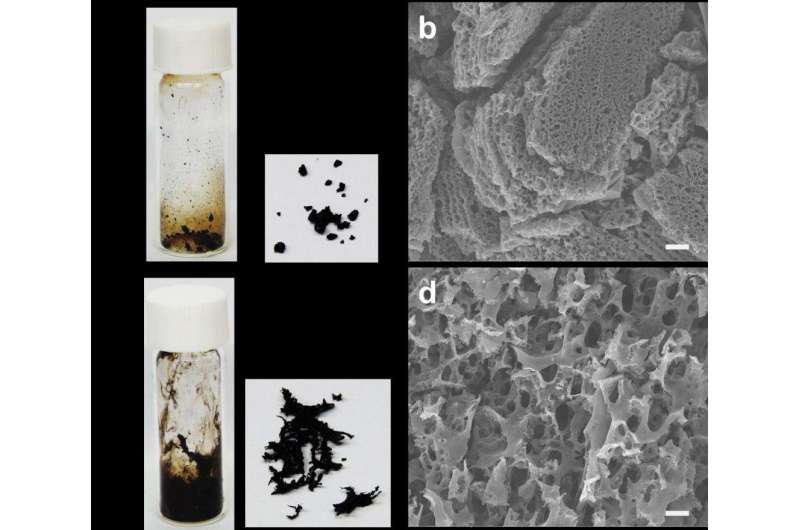
"Solid-state reactions can yield products of high purity and therefore post-treatment for purification may become unnecessary," says Dr. Seo-Yoon Bae in the School of Energy and Chemical Engineering, the first author of the study. "Additionally, the resultant products are of high purity, and therefore, have more advantages than solution or gas-phase reactions."

The HEA single crystals consists of nine HEA molecules with two acetone and one water molecules in the lattice. High exothermic heat is explosively released because the boiling point of water and acetone is low, according to the research team. Since water boils at 100°C and acetone boils at 56°C, there is an increase in the kinetic energies between the two molecules when heat is applied. Thus, before the organic single crystals melt, the acetone and water molecules (primer to trigger explosion) are released outside, HEA molecules in the crystal lattice start rearranging and this is followed by high exothermic heat (gunpowder explosion).
More information: Seo-Yoon Bae et al, Forming a three-dimensional porous organic network via solid-state explosion of organic single crystals, Nature Communications (2017). DOI: 10.1038/s41467-017-01568-3
Journal information: Nature Communications




















