Green functionalisation of carbon-hydrogen bonds

NUS chemists have developed a light enhanced, nickel catalysed method for carbon-hydrogen (C-H) bond functionalisation in organic molecules.
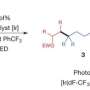
The replacement of "H" in C–H bonds with other atoms or substituents is one of the most coveted ways to create new and useful molecules. However, while the C-H bond can be found in most organic molecules, it is considered inactive and therefore difficult to functionalise. C-H bond functionalisation refers to the substitution of the hydrogen atom in the C-H bond with another functional group, such as an alkene, alkyne, amine, alcohol, ester, aldehyde, acid, etc. Prof WU Jie and his research group from the Department of Chemistry, NUS have developed an environmentally friendly process that uses visible light and nickel catalysts to enable the vinylation of ethers and amides via C-H bonds. Their method does not require special chemicals or other additives which might result in the generation of hazardous substances.
Ether and amide chemical groups are present in many pharmaceutically active agents. This new method provides an environmentally friendly way for pharmaceutical companies to produce pharmaceutical products involving the modification of these chemical groups.
Prof Wu said, "The products formed from this new process cannot be obtained directly through other synthetic means using alkynes as the starting material. We also discovered that the hydrogen atom in the C-H bond detaches and binds itself onto the nickel catalyst, becoming a nickel-hydride intermediary during the reaction. The hydrogen atom is then released back into the resultant molecule. This type of C-H bond activation mechanism is rarely seen in organic reactions and further research can be explored to improve our understanding of it."
The research team plans to continue developing better visible light promoted chemical transformations to enable direct C-H bond functionalisations for organic compounds which are useful for the pharmaceutical industry.
More information: Hong-Ping Deng et al. Photoinduced Nickel-Catalyzed Chemo- and Regioselective Hydroalkylation of Internal Alkynes with Ether and Amide α-Hetero C(sp3)–H Bonds, Journal of the American Chemical Society (2017). DOI: 10.1021/jacs.7b08158
Journal information: Journal of the American Chemical Society
Provided by National University of Singapore