Study reveals unknown details about common lithium-ion battery materials
High-performance electrodes for lithium-ion batteries can be improved by paying closer attention to their defects—and capitalizing on them, according to Rice University scientists.
Rice materials scientist Ming Tang and chemists Song Jin at the University of Wisconsin-Madison and Linsen Li at Wisconsin and the Massachusetts Institute of Technology led a study that combined state-of-the-art, in situ X-ray spectroscopy and modeling to gain insight into lithium transport in battery cathodes. They found that a common cathode material for lithium-ion batteries, olivine lithium iron phosphate, releases or takes in lithium ions through a much larger surface area than previously thought.
"We know this material works very well but there's still much debate about why," Tang said. "In many aspects, this material isn't supposed to be so good, but somehow it exceeds people's expectations."
Part of the reason, Tang said, comes from point defects—atoms misplaced in the crystal lattice—known as antisite defects. Such defects are impossible to completely eliminate in the fabrication process. As it turns out, he said, they make real-world electrode materials behave very differently from perfect crystals.
That and other revelations in a Nature Communications paper could potentially help manufacturers develop better lithium-ion batteries that power electronic devices worldwide.
The lead authors of the study—Liang Hong of Rice and Li of Wisconsin and MIT—and their colleagues collaborated with Department of Energy scientists at Brookhaven National Laboratory to use its powerful synchrotron light sources and observe in real time what happens inside the battery material when it is being charged. They also employed computer simulations to explain their observations.

One revelation, Tang said, was that microscopic defects in electrodes are a feature, not a bug.
"People usually think defects are a bad thing for battery materials, that they destroy properties and performance," he said. "With the increasing amount of evidence, we realized that having a suitable amount of point defects can actually be a good thing."
Inside a defect-free, perfect crystal lattice of a lithium iron phosphate cathode, lithium can only move in one direction, Tang said. Because of this, it is believed the lithium intercalation reaction can happen over only a fraction of the particle's surface area.
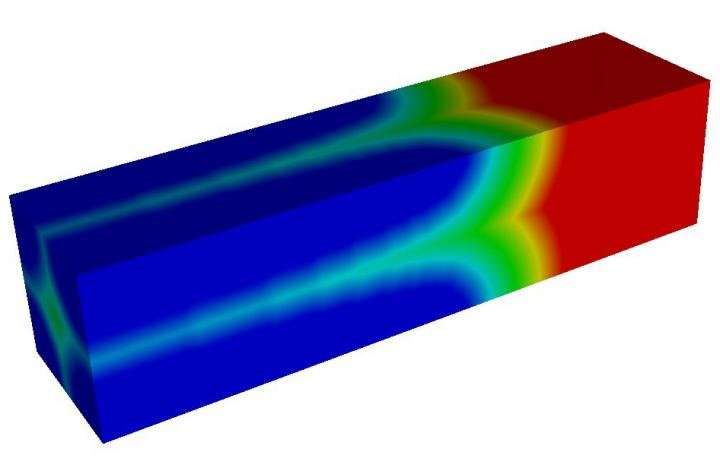
But the team made a surprising discovery when analyzing Li's X-ray spectroscopic images: The surface reaction takes place on the large side of his imperfect, synthesized microrods, which counters theoretical predictions that the sides would be inactive because they are parallel to the perceived movement of lithium.
The researchers explained that particle defects fundamentally change the electrode's lithium transport properties and enable lithium to hop inside the cathode along more than one direction. That increases the reactive surface area and allows for more efficient exchange of lithium ions between the cathode and electrolyte.
Because the cathode in this study was made by a typical synthesis method, Tang said, the finding is highly relevant to practical applications.

"What we learned changes the thinking on how the shape of lithium iron phosphate particles should be optimized," he said. "Assuming one-dimensional lithium movement, people tend to believe the ideal particle shape should be a thin plate because it reduces the distance lithium needs to travel in that direction and maximizes the reactive surface area at the same time. But as we now know that lithium can move in multiple directions, thanks to defects, the design criteria to maximize performance will certainly look quite different."
The second surprising observation, Tang said, has to do with the movement of phase boundaries in the cathode as it is charged and discharged.
"When you take heat out of water, it turns into ice," he said. "And when you take lithium out of these particles, it forms a different lithium-poor phase, like ice, that coexists with the initial lithium-rich phase." The phases are separated by an interface, or a phase boundary. How fast the lithium can be extracted depends on how fast the phase boundary moves across a particle, he said.
Unlike in bulk materials, Tang explained, it has been predicted that phase boundary movement in small battery particles can be limited by the surface reaction rate. The researchers were able to provide the first concrete evidence for this surface reaction-controlled mechanism, but with a twist.
"We see the phase boundary move in two different directions through two different mechanisms, either controlled by surface reaction or lithium bulk diffusion," he said. "This hybrid mechanism paints a more complicated picture about how phase transformation happens in battery materials. Because it can take place in a large group of electrode materials, this discovery is fundamental for understanding battery performance and highlights the importance of improving the surface reaction rate."
More information: Liang Hong et al, Two-dimensional lithium diffusion behavior and probable hybrid phase transformation kinetics in olivine lithium iron phosphate, Nature Communications (2017). DOI: 10.1038/s41467-017-01315-8
Journal information: Nature Communications
Provided by Rice University