Nice ice, maybe: Study finds water-repelling surfaces ease ice removal
Water-repellent surfaces and coatings could make ice removal a literal breeze by forcing ice to grow up rather than just skate by, says a new study from the University of Nebraska-Lincoln and several Chinese institutions.
The researchers discovered that ice grows differently on absorbent vs. water-repellent surfaces, demonstrating that a gust of air can blow away ice that forms on the latter. Their findings suggest that applying water-repellent coatings to windshields before winter storms - or engineering surfaces that inherently repel water - could enable a strong breeze to handle the burden of ice removal.
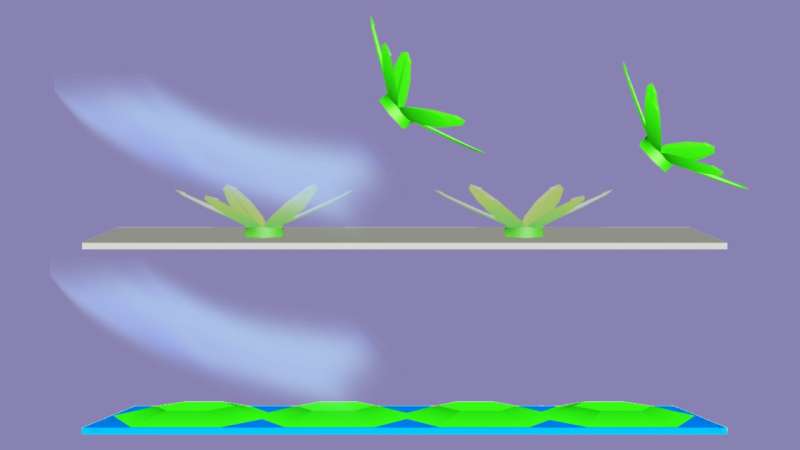
Experiments and simulations showed that a water droplet on a repellent surface will freeze upward into a microscopic six-armed formation that resembles an idealized snowflake, with only a small portion of its base adhering to the surface. This makes sense given that water droplets bead up rather than spread out over repellent surfaces, said Nebraska co-author Xiao Cheng Zeng.
In contrast, droplets on an absorbent surface crystallized into ice that grew along that surface, making it more difficult to remove. Molecular-level simulations suggested that these droplets almost immediately began forming two stacked layers of hexagonal 2-D ice, a form that Zeng previously discovered and dubbed Nebraska Ice. This ultra-thin ice encourages water molecules to essentially skate across it and colonize other areas of the surface, Zeng said.
"If the water and the surface don't have much chemistry in the beginning - they don't like each other - it's kind of like a divorce or separation," said Zeng, Chancellor's University Professor of chemistry. "But if they like each other, they marry and stay together for a long time.
"That's when the ice grows along the surface. In the winter, if you have that kind of ice on a windshield, you have to use a scraper to get it off."
Onward or upward
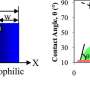
Temperature and pressure mostly dictate how water droplets crystallize in open air, and those variables do factor into ice formation on solid surfaces, Zeng said. But the team's study suggests that a surface's contact angle - the angle formed where a water droplet meets a solid surface - determines whether ice will grow along or off the surface. Whereas a hydrophilic surface allows droplets to spread across it at a small contact angle, a water-repelling hydrophobic surface will force droplets to bead up and form a larger angle.
"Whether water freezes in one way or the other is up to the surface, not the temperature," Zeng said. "It's almost entirely dependent on the contact angle."
On a defect-free surface fabricated in the lab or modeled in a computer simulation, ice transitions from along-surface to off-surface growth at a contact angle of somewhere between 30 and 40 degrees, the team found. The researchers also discovered that increasing the roughness of a surface by enlarging its nanoscopic pores actually decreased this angular threshold, meaning that rougher surfaces need not be as water-repellent to foster the growth of more-easily removed ice.
Breaking the ice
To compare the two forms of ice growth, the researchers designed a transparent surface split into halves: one hydrophilic, one hydrophobic. They then attached a high-speed camera to a microscope, capturing video of the respective processes both from beneath and from a side profile.
When the researchers subjected both halves to puffs of air, they found that ice abandoned the hydrophobic half but steadfastly held to the hydrophilic side. And ice that advanced across the hydrophilic half abruptly halted when it neared hydrophobic territory.
"People have been studying how water interacts with surfaces for a long, long time," Zeng said. "But this phenomenon was off the radar until now."
Zeng authored the study with Nebraska's Chongqin Zhu, postdoctoral researcher in chemistry; Joseph Francisco, dean of the College of Arts and Sciences; along with colleagues from the Chinese Academy of Sciences, Beijing University of Chemical Technology, and Peking University. The team reported its findings in the journal Proceedings of the National Academy of Sciences.
More information: Jie Liu et al, Distinct ice patterns on solid surfaces with various wettabilities, Proceedings of the National Academy of Sciences (2017). DOI: 10.1073/pnas.1712829114
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Nebraska-Lincoln