Even droplets sometimes take the stairs
Sometimes, liquid drops don't drop. Instead, they climb. Using computer simulations, researchers have now shown how to induce droplets to climb stairs all by themselves.
This stair-climbing behavior could be useful in everything from water treatment and new lab-on-a-chip microfluidic devices, to biochemical processing and medical diagnostic tools. The researchers, from the Indian Institute of Technology in Roorkee, India, and York University in Toronto, describe their findings this week in the journal Physics of Fluids.
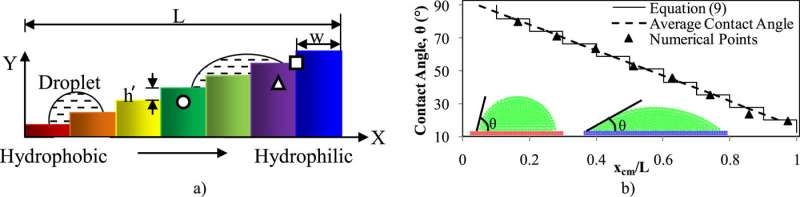
To get the droplets to climb, this new research reveals you need a staircase whose surface adheres to each droplet more readily with each step. A surface on which a droplet sticks easily has what's called a high wettability, causing the droplet to spread out and flatten. On a low-wettability surface, however, the droplet would stay more spherical, like raindrops beading up on a waterproof jacket.
The researchers have previously used a gradient of increasing wettability to coax droplets to move across a flat surface and even to go up a slope. A water droplet, for example, is more attracted to a hydrophilic surface with its greater wettability, so an incline featuring an increasing hydrophilic surface as it rises can "pull" a droplet uphill.
Real surfaces are never perfectly smooth, however; at small-enough scales, a surface eventually appears rough. A slope at these scales is actually a microscopic staircase. "Most surfaces are textured, and mobility of a droplet over such surfaces require climbing stairs," said Arup Kumar Das of IIT Roorkee.
To explore how a droplet could climb steps—and thus if this technique can work on more real-world surface applications—the researchers simulated the physics of microliter-sized droplets on staircases with a wettability gradient.
These droplets are wider than the length of each step, so their leading side is on a higher step with a more wettable surface, than the trailing side. The front part of the droplet thus spreads more, forming a smaller, flatter angle with the surface.
The difference in angles between the front and back of the climbing droplets causes the liquid inside the droplet to circulate. When the leading edge of the droplet reaches the next step, the circulation drives the droplet forward, spilling over onto the next higher step, and the process repeats itself.
Whether the droplet has enough force to overcome gravity depends on the size of the droplet, the steepness of the steps and the differences in wettability. In general, a bigger droplet is better at climbing stairs, and for steeper steps, there needs to be a higher wettability gradient.
The researchers are now working on experiments to confirm the simulation results.
Many other methods to control droplets rely on external forces such as temperature variations, and electric and magnetic fields. But, Das explained, those methods are often challenging and complex. The new study shows that passive approaches like wettability could be more efficient. "Passive means [we] can manipulate a droplet to even climb stairs sustainably without using an external force," he said.
More information: Prabh P. S. Seerha et al, Proposition of stair climb of a drop using chemical wettability gradient, Physics of Fluids (2017). DOI: 10.1063/1.4985213
Journal information: Physics of Fluids
Provided by American Institute of Physics




















