Molecular 'samurai sword' controls plant growth

Researchers from AMOLF and Wageningen University & Research have used a smart simulation program to study a plant protein called katanin and published their findings in the journal PNAS.
Katanin, named for Japanese katana swords, lives up to its name. The protein severs the protein-based microtubules that serve as a scaffold for the basic building blocks of plants such as cellulose. As a result, the microtubules grow in the right direction, guide further growth and division of the plant cells, and consequently the development of the entire plant.
Extending microtubules
Microtubules can grow up to several tens of micrometers in length. They are located along the inside of the plant cell membrane and stretch out along the inside of the cell. The growth and division of a plant cell occurs perpendicular to the direction in which the microtubules extend. "You can compare the cortical microtubule array with a corset that you can unfold," says Eva Deinum, first author of the publication and university lecturer at Wageningen University & Research (WUR). "Internally, plant cells are under considerable pressure, as a result of which they want to expand, grow and divide in all directions. The array determines in which direction that happens."
Self-organizing mechanism
Plant cells are incredibly large by comparison with animals cells, in which a central distribution point for microtubules suffices. Plants, however, need a decentralized and self-organizing mechanism that carefully manages the growth of the microtubules. The "samurai sword" protein katanin was found to play a crucial role in this mechanism.

Not indiscriminate severing
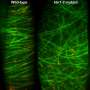
For a long time, it was thought that katanin gave rise to more chaos, because the number of degrees of freedom increases if the microtubules are severed in two. This conclusion proved to be shortsighted, however. The researchers, under the leadership of Bela Mulder, discovered that the protein does not sever indiscriminately. It works according to a fixed strategy that guarantees all of the microtubules grow in the right direction.
"The microtubules are programmed in such a way that if they collide with each other – and therefore come into contact with each other – they stop growing and even start to shrink," says Mulder. "That is the basic mechanism underlying the organization. The katanin comes into action as a backup to this basic mechanism if two microtubules cross each other without colliding—in other words, if they do not grow in the same direction. Here, we can see that the protein usually severs the microtubule lying on top, i.e., the tube that arrived last."
With these observations, the researchers have solved the katanin paradox. They have demonstrated that katanin guides a selection mechanism that promotes the organization of the microtubules – the corset in which they are packed together.
'In silico' tests
A vital aspect of the research was the simulation program that Mulder's group used. This was originally developed by his former PhD student Simon Tindemans and subsequently expanded and refined by Eva Deinum during her doctoral research at AMOLF. Using that program, the researchers could observe how small variations in the katanin protein led to radically different severing behavior, which resulted in complete chaos. In the simulations, the group performed 'in silico' tests of the effects of various mutations on the protein's function. This revealed that only the natural, non-mutated variant possessed the correct severing strategy.
The high simulation speed was crucial for the success of the research, says Deinum. "As a result of this, we could run simulations 10,000 times faster than those of competing research groups. We not only worked far faster, but we could also test more variations of external parameters and conditions. This made a qualitative difference to the results we obtained, as well. For example, we could accurately determine the conditions under which the kantanin protein promoted the ordering."
More information: How selective severing by katanin promotes order in the plant cortical microtubule array, Eva E. Deinum, Simon H. Tindemans, Jelmer J. Lindeboom, en Bela M. Mulder, PNAS, DOI: 10.1073/pnas.1702650114
Journal information: Proceedings of the National Academy of Sciences
Provided by AMOLF