Scientists develop new concept of confined catalysis under 2-D materials
Small spaces in nanoreactors may have big implications for chemistry. The chemical nature of molecules and reactions within nanospaces can be changed significantly due to the nanoconfinement effect. Understanding the fundamentals of confined catalysis has become an important topic in heterogeneous catalysis. 2-D nanoreactors formed under 2-D materials can provide a well-defined model for exploring confined catalysis.
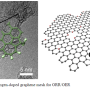
A research group led by Profs. FU Qiang and BAO Xinhe of the Chinese Academy of Sciences has published a study in PNAS revealing the geometric constraint and confinement field in two-dimensional (2-D) space between a graphene overlayer and Pt(111). The study demonstrates a new concept of confined catalysis under 2-D materials, which they have named "catalysis under cover."
The scientists chose a graphene/Pt (111) surface as a model for studying confined catalysis using density functional theory (DFT) calculations. They showed that the adsorption of atoms and molecules on the Pt(111) surface is weakened under graphene. A similar result has been found on Pt(110) and Pt(100) surfaces covered with graphene. Both the geometric constraint and confinement field imposed by the 2-D cover are attributed to the observed confinement phenomena.
The general tendency for weakened surface adsorption under the confinement of a graphene overlayer enables feasible modulation of surface reactions by placement of a 2-D cover. The concept "catalysis under cover" can be applied to reactions between two opposite 2-D walls interacting with each other through van der Waals forces. The concept helps in the design of high-performance nanocatalysts interfacing with 2-D material overlayers.
The research group demonstrated the confinement-induced modulation of surface reactivity in a Pt-catalyzed oxygen reduction reaction (ORR) under 2-D covers. It is known that oxygen binding to Pt is relatively strong, and all means of weakening this binding can be used to promote the reaction. When placing different 2-D materials such as graphene and h-BN on the surface, oxygen binding with Pt weakens, thus effectively enhancing ORR activity.
Confined catalysis under 2-D materials can be applied to supported nanocatalysts. Metal nanoparticles may be encapsulated by 2-D materials, thus forming core-shell nanostructures. The active core structures are well protected by the outer shells and catalyst stability is improved. Furthermore, catalyst activity can be enhanced by the confinement of the outer shells.
More information: Haobo Li et al, Confined catalysis under two-dimensional materials, Proceedings of the National Academy of Sciences (2017). DOI: 10.1073/pnas.1701280114
Journal information: Proceedings of the National Academy of Sciences
Provided by Chinese Academy of Sciences