Promising graphene catalyst obtained from sticky rice
Recently, researchers from Tsinghua University, Queen Mary University of London, and Institute of Metal Research, Chinese Academy of Sciences, have reported a promising graphene catalyst obtained from sticky rice, and revealed the critical importance of topological defects both experimentally and theoretically.
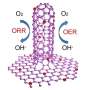
"Oxygen reduction/evolution reaction (ORR/OER) electrocatalysis is the core issue of some next-generation energy systems, such as fuel cells, metal-air batteries and so on," said Prof. Qiang Zhang from the Department of Chemical Engineering, Tsinghua University. "Metal-free nanocarbon materials have been intensively investigated as ORR/OER catalysts, due to their remarkable activity, high conductivity and flexibility, the tunable structure and surface chemistry, facile preparation, and economic viability."
However, up to now, the exact active sites in nanocarbon materials and their specific roles during ORR and OER remain elusive and controversial. Additionally, affordable and scalable methods to obtain excellent catalysts are still required for practical application.
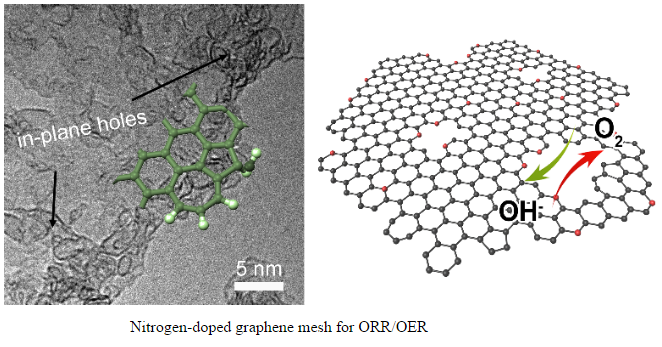
"We proposed a novel graphene mesh with tunable nitrogen doping and topological defects, and fabricated via the direct carbonization of a ternary composite with sticky rice, melamine, and Mg(OH)2 templates." said Cheng Tang, a Ph.D. student from Tsinghua University and the first author. "Then unexpectedly, an excellent ORR performance superior to Pt/C in terms of both activity and stability, and OER activity comparable with Ir/C is obtained with a low overpotential gap of 0.90 V, making it one of the best bifunctional, metal-free catalysts ever reported."
Compared with other samples, they found that the topological defects in graphene edges seem to be more important than the nitrogen doping for higher activity. "It is different from previous results, but important for a thorough understanding of the activity origins of nanocarbon catalysts," said Prof. Zhang.
Density function theory calculations were conducted to elucidate the experimental results. N-doped moieties are demonstrated to present a higher overpotential than pristine graphene nanoribbons, but those moieties with five-carbon or seven-carbon ring defects exhibit enhanced activity. A nitrogen-free configuration with adjacent pentagon and heptagon carbon rings is found to exhibit the lowest overpotential for both ORR and OER.
"This work provides fresh insights into the origins of oxygen electrocatalytic activity for metal-free nanocarbon electrocatalysts," C. Tang explained. "The importance of topological defects, in addition to the doping-induced sites in the heteroatom-doped nanocarbon materials is elucidated towards promising ORR/OER catalysis."
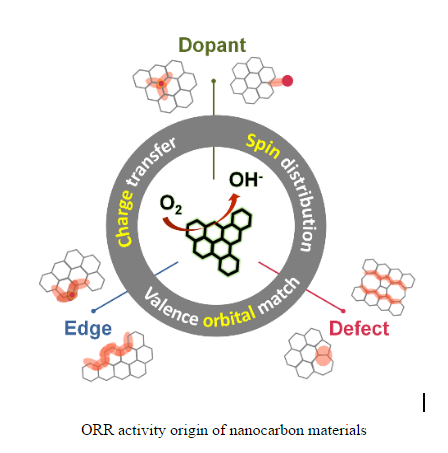
After systematical comparison of previous reports, Prof. Zhang and co-workers proposed that different active sites, such as dopants, edges and defects, actually generate promising ORR activity to metal-free nanocarbon materials by the analogous modification of electron structures, leading to optimized intermediate chemisorption and facilitated electron transfer.
Such syncretic understanding should contribute to further research. "We believe that specific heteroatom doping at defective edges most effectively alter electron structures and achieve optimal ORR activities for metal-free electrocatalysts."
More information: Cheng Tang et al. Topological Defects in Metal-Free Nanocarbon for Oxygen Electrocatalysis, Advanced Materials (2016). DOI: 10.1002/adma.201601406
Cheng Tang et al. Nanocarbon for Oxygen Reduction Electrocatalysis: Dopants, Edges, and Defects, Advanced Materials (2017). DOI: 10.1002/adma.201604103
Journal information: Advanced Materials
Provided by Tsinghua University