May 10, 2017 report
Researchers isolate hydrogen fluoride and water to understand acid dissolution
(Phys.org)—Beginning level chemistry classes learn about Brønsted-Lowry acids. These acids dissociate in water to form an H+ ion and a negatively charged counterion. While this is a fundamental lesson, the actual mechanism of dissolution is a bit of a mystery. The H+ molecule combines with water to form H3O+, but the number of water molecules needed to hydrate the simplest acid, HF, is unknown. Attempts to isolate HF and H2O are difficult, largely because of the high reactivity of HF and the tendency of water to form hydrogen bonds.
To understand the fundamental mechanism behind acid dissolution, Zhang et al. from the Institute for Chemical Research at Kyoto University encapsulated HF, as well as HF•H2O and H2O within a C70 fullerene. They found that in order to force the molecules into the open fullerene cavity, the molecules required "pushing from the outside" using high pressure conditions, and "pulling from the inside" via molecular interactions between HF and H2O. They were able to identify how hydrogen bonding occurred between these two molecules. Their work appears in Science Advances.
Prior work by Zhang et al. showed that the C70 fullerene could be opened in a three-step process that involved the addition of a pyridazine derivative either to the alpha or beta bonds on the C70. This created a 13-member ring opening that formed slightly different compounds, denoted by α-13mem and β-13mem. Dehydration of both compounds resulted in a 16-member ring opening. The ring could be closed again via hydrolysis and a two-step process.
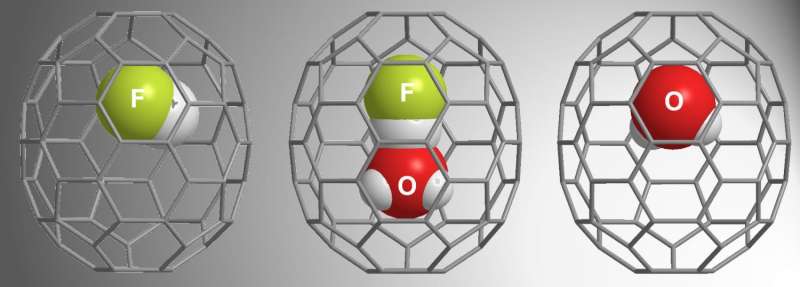
β-16mem was large enough to capture H2O, but α-16mem was not. Given these results from previous studies, for the current study, Zhang et al. used α-16mem to try to encapsulate HF. Instead, they found three different possibilities within the fullerenes: HF@C70, (HF•H2O)@C70, and H2O@C70.
Their reaction conditions required high pressure (9000 atm) to "push" the guest molecule into the α-16mem cavity. Time-dependent studies showed that HF filled the cavity first, followed by H2O•HF, and then H2O. Notably, the open cage did not entrap H2O when HF was not present, indicating that the interaction between H2O and HF prompted H2O encapsulation. Further studies showed that HF is "pulling" H2O into the cavity while the high pressure environment "pushes" it into the cavity.
This process allowed the authors to study the interaction between H2O and HF within a confined environment using 1H NMR. NMR analysis showed that that the (H2O•HF)@C70 was down-shifted from H2O@C70 and HF@C70, which indicated hydrogen bonding. Furthermore, shift and coupling values indicated that oxygen was acting as the hydrogen-bond acceptor.
Using single-crystal x-ray diffraction, Zhang et al. demonstrated the structure of the (HF•H2O)@C70, and report the first x-ray structure for doubly encapsulated C70. These analyses and experimental studies confirmed that the H+ ion in HF forms a linear hydrogen bond with the O in H2O. Additionally, compared to theoretical calculations of free H2O and HF, the studies of the encapsulated molecules revealed close contact with hydrogen and oxygen that may be characteristic of H3O+•F-.
The C70 fullerene derivative provides an excellent nanoenvironment for studying isolated chemical species, something that has not been available to chemists in the past. This isolated environment allowed the authors to investigate the interactions of two compounds without interference from the surrounding environment and provided important insights into a ubiquitous chemical process.
More information: Rui Zhang et al. Isolation of the simplest hydrated acid, Science Advances (2017). DOI: 10.1126/sciadv.1602833
Abstract
Dissociation of an acid molecule in aqueous media is one of the most fundamental solvation processes but its details remain poorly understood at the distinct molecular level. Conducting high-pressure treatments of an open-cage fullerene C70 derivative with hydrogen fluoride (HF) in the presence of H2O, we achieved an unprecedented encapsulation of H2O·HF and H2O. Restoration of the opening yielded the endohedral C70s, that is, (H2O·HF)@C70, H2O@C70, and HF@C70 in macroscopic scales. Putting an H2O·HF complex into the fullerene cage was a crucial step, and it would proceed by the synergistic effects of "pushing from outside" and "pulling from inside." The structure of the H2O·HF was unambiguously determined by single crystal x-ray diffraction analysis. The nuclear magnetic resonance measurements revealed the formation of a hydrogen bond between the H2O and HF molecules without proton transfer even at 140°C.
Journal information: Science Advances
© 2017 Phys.org