April 21, 2017 report
X-ray scattering and molecular dynamics simulations used to create real-time imaging of nanoparticle oxidation
(Phys.org)—A team of researchers affiliated with Temple University and Argonne National Laboratory has developed a way to observe material restructuring at the atomic scale in real time. In their paper published in the journal Science, the team describes their technique and what they observed as they followed the progression of oxidation at the atomic scale. Doris Cadavid and Andreu Cabot with Catalonia Institute for Energy Research offer a Perspective piece on the work done by the team in the same journal issue, and also outline the history and difficulties associated with observing atomic-level changes occurring in a material. They note also that the newly developed technique is likely to have a major impact on how metals and other compounds are engineered in the future.
Humans have known for thousands of years, as Cadavid and Cabot note, that materials decompose, burn or rust, and have learned more recently that such changes occur at the atomic level. To learn more about such processes, scientists have studied them in depth, but have been partially limited by an inability to actually watch what happens at the atomic level. That may be changing, as the researchers with this new effort have developed a way to watch oxidation occurring at the atomic level in real time.
The method involved combining a small-angling X-ray scattering technique with molecular modeling software to track in precise detail the oxidation process of iron oxide nanoparticles—all in real time.
The technique allowed the researchers to see that empty spaces would form at the onset of the process, which fused together when they grew to a certain size, creating other larger crescent-shaped empty spaces. They also found that they could control the diffusion process with the empty spaces by altering the temperature and size of the nanoparticles.
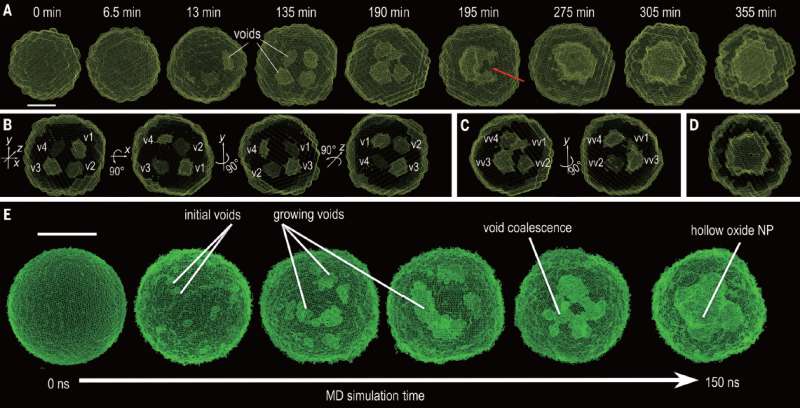
Cadavid and Cabot suggest that the technique may herald the dawn of a new era in chemistry—the ability to watch the process of solids being modified at the atomic scale in real time, or slowed down for fast reactions. It could lead, they suggest further, to better controlling such processes, including finding new ways to prevent metals from suffering damage due to rusting.
More information: Yugang Sun et al. Quantitative 3D evolution of colloidal nanoparticle oxidation in solution, Science (2017). DOI: 10.1126/science.aaf6792
Abstract
Real-time tracking of the three-dimensional (3D) evolution of colloidal nanoparticles in solution is essential for understanding complex mechanisms involved in nanoparticle growth and transformation. We used time-resolved small-angle and wide-angle x-ray scattering simultaneously to monitor oxidation of highly uniform colloidal iron nanoparticles, enabling the reconstruction of intermediate 3D morphologies of the nanoparticles with a spatial resolution of ~5 angstroms. The in situ observations, combined with large-scale reactive molecular dynamics simulations, reveal the details of the transformation from solid metal nanoparticles to hollow metal oxide nanoshells via a nanoscale Kirkendall process—for example, coalescence of voids as they grow and reversal of mass diffusion direction depending on crystallinity. Our results highlight the complex interplay between defect chemistry and defect dynamics in determining nanoparticle transformation and formation.
Journal information: Science
© 2017 Phys.org