Plant-made virus shells could deliver drugs directly to cancer cells
Viruses are extremely efficient at targeting and delivering cargo to cells. In the journal ACS Nano, researchers report they have harnessed this well-honed ability—minus the part that makes us sick—to develop virus-like nanoparticles to deliver drugs straight to affected cells. In lab tests, they show that one such particle can be produced in plants and it ferries small molecules to cancer cells.
For this work, Frank Sainsbury and colleagues copied the core protein shell of the Bluetongue virus, a pathogen that affects ruminant animals. Previous research has shown that the capsid is stable, has a large cavity for small molecules or proteins to pack into, and is easy to produce with high purity. The researchers wanted to try making the virus-shell nanoparticles using plants. This is an increasingly popular approach to producing pharmaceuticals as it minimizes possible contamination by human pathogens, which plants don't carry. But first they needed to understand the structure of the shells.
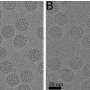
Using single particle cryo-electron microscopy, the team showed for the first time that the recombinant shell nanoparticles produced by plants were different from the natural virus capsid. With the nanoparticles' detailed structure in hand, the researchers then genetically and chemically engineered them to their specifications, and loaded proteins and small molecules inside the shells. Lab testing showed that the plant-made virus particles, which naturally bind to receptors on cancer cells, were taken in by human breast cancer cells. The findings suggest the nanoparticles can potentially be used for the targeted delivery of drugs.
More information: Engineering Recombinant Virus-like Nanoparticles from Plants for Cellular Delivery, ACS Nano, Article ASAP, DOI: 10.1021/acsnano.6b07747
Abstract
Understanding capsid assembly following recombinant expression of viral structural proteins is critical to the design and modification of virus-like nanoparticles for biomedical and nanotechnology applications. Here, we use plant-based transient expression of the Bluetongue virus (BTV) structural proteins, VP3 and VP7, to obtain high yields of empty and green fluorescent protein (GFP)-encapsidating core-like particles (CLPs) from leaves. Single-particle cryo-electron microscopy of both types of particles revealed considerable differences in CLP structure compared to the crystal structure of infection-derived CLPs; in contrast, the two recombinant CLPs have an identical external structure. Using this insight, we exploited the unencumbered pore at the 5-fold axis of symmetry and the absence of encapsidated RNA to label the interior of empty CLPs with a fluorescent bioconjugate. CLPs containing 120 GFP molecules and those containing approximately 150 dye molecules were both shown to bind human integrin via a naturally occurring Arg-Gly-Asp motif found on an exposed loop of the VP7 trimeric spike. Furthermore, fluorescently labeled CLPs were shown to interact with a cell line overexpressing the surface receptor. Thus, BTV CLPs present themselves as a useful tool in targeted cargo delivery. These results highlight the importance of detailed structural analysis of VNPs in validating their molecular organization and the value of such analyses in aiding their design and further modification.
Journal information: ACS Nano
Provided by American Chemical Society