Physical basis of tissue coordination uncovered
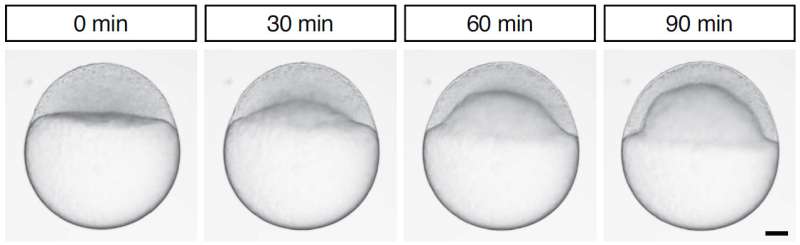
The little striped zebrafish starts out as single big cell sitting on top of the yolk. During the next 3 days, cells divide and tissues move to give the fish its final shape. But how do tissues coordinate their often-complicated movements? The physical basis of tissue coordination in early zebrafish development is subject of a study by Carl-Philipp Heisenberg, Professor at the Institute of Science and Technology Austria (IST Austria) and his group, including first author and postdoc Hitoshi Morita, and colleagues at The Francis Crick Institute in London and the Max-Planck-Institute for the Physics of Complex Systems in Dresden. Until now, little has been known about how tissues coordinate their movement both temporally and spatially during development.
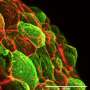
In the study, published today in Developmental Cell, Heisenberg and co-authors investigated how tissues coordinate their movements and how the forces required for tissue movements are generated. In the paper, they show that cells at the surface are crucial for coordination. Carl-Philipp Heisenberg explains: "A reduction of surface tension by cells on the surface of the embryo is the key process that coordinates tissue movements at this timepoint."
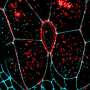
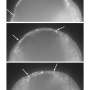
Tissue spreading is a key process both in development and disease, for example in wound healing. For a tissue, especially a complex tissue with several layers, to spread, it needs to simultaneously thin and expand. One example of such spreading is the so-called doming in the zebrafish embryo. During doming, the blastoderm, a tissue composed of surface epithelial cells and inner mesenchymal cells, thins and spreads over the yolk cell. Doming involves two tissue movements: the epithelial cell layer at the surface expands, and inner cells undergo intercalations thereby thinning and spreading the inner cell mass. In the present study, Heisenberg and his colleague asked how the two tissue movements - surface cell expansion and inner cell intercalation - coordinate their movements during blastoderm spreading.
Combining theory and experiments, they show that surface cells, by undergoing active expansion, reduce the surface tension of the blastoderm. Strikingly, this loosening at the blastoderm surface not only triggers surface cell layer expansion, but also induces inner cell intercalation leading to inner cell layer thinning and spreading. Thus, the reduction in blastoderm surface tension represents the key process coordinating surface cell layer expansion with inner cell layer thinning and spreading during doming. First author Hitoshi Morita explains the significance of this study for understanding tissue spreading: "We have unravelled the force-generating processes that drive doming. Our study shows that by reducing its surface tension, the layer of epithelial cells simultaneously drives expansion and thinning of the blastoderm, and so coordinates these two processes. Coordinated tissue spreading is a universal mechanism by which embryos take shape. Understanding the force-generating mechanism is central for understanding the physical basis of embryo development. We have uncovered the key role surface cells play in this process."
More information: Developmental Cell, DOI: 10.1016/j.devcel.2017.01.010
Journal information: Developmental Cell
Provided by Institute of Science and Technology Austria