Materials made of self-spinning particles
Matter is either gas, liquid or solid based on how its molecules respond to temperature and pressure. But what if the building blocks are self-spinning particles instead of ordinary molecules? Theoretical physicists found out what determines the phase of those artificial materials. The research has been reported in PNAS.
When water reaches 100 °C, it transitions to a gas phase, at least at sea level. Removing air pressure causes water to boil at colder temperatures. It is clear that materials made up of ordinary molecules take on a phase depending on temperature and pressure. Leiden theoretical physicist Prof. Vincenzo Vitelli wondered what would happen if materials had self-spinning dimers as building blocks instead.
Simulation
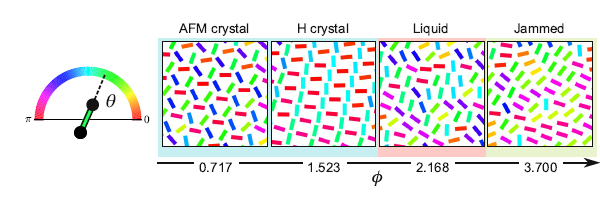
To this end, first authors Benny van Zuiden and Jayson Paulose simulated self-spinning dimers in silico and studied how they organize themselves. By applying a gradually increasing pressure on them, they saw the system change from an ordered state to a very chaotic state.
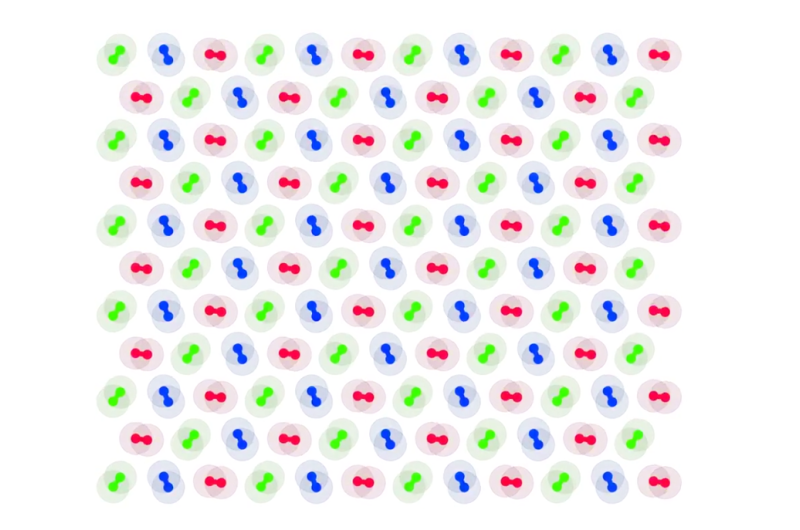
The figure below (left) shows a beautifully ordered state, with dimers neatly forming a triangular crystal lattice. Moreover, the relative orientation of nearby particles are locked as they spin.
At right, the concentration is so high that the system gets stuck in a glassy phase. Remarkably, there is a liquid phase in between. Usually, a substance becomes more solid as its density increases. Here, the opposite happens.
Liquid
So how can there be a liquid state? With low density, the dimers have plenty of room to move as they wish and stay in sync, like a group of stage dancers. When the stage is too small, dancers will bump in to each other and move chaotically, as particles in a liquid. However, if the stage gets so tiny that the dancers are unable to move, they get stuck in a disordered configuration reminiscent of a glass.
More information: Benjamin C. van Zuiden et al. Spatiotemporal order and emergent edge currents in active spinner materials, Proceedings of the National Academy of Sciences (2016). DOI: 10.1073/pnas.1609572113
Journal information: Proceedings of the National Academy of Sciences
Provided by Leiden Institute of Physics