Sodium-oxygen batteries have improved cycle life due to highly concentrated electrolytes
Smartphones, laptops, electric cars—whatever the device, an efficient battery is high on any user's wish list. The search for the next-generation battery has recently focused on sodium– oxygen batteries. Theoretically, these should provide previously unattainable efficiency but their practical implementation has proven to be a stumbling block. Researchers now report in the journal Angewandte Chemie, that a highly concentrated electrolyte solution may make the sodium–oxygen battery more stable, and therefore more practicable.
Researchers have high hopes for alkali metal/oxygen batteries, because their theoretical energy density is particularly high. In such batteries, one electrode is made from the pure alkali metal. Upon discharging, this electrode gives up electrons to the circuit and positive ions to the electrolyte. The counter electrode is made of porous carbon and is in contact with the air. At this electrode, oxygen is reduced by taking up electrons in the presence of the metal ions. This may result in a variety of metal oxide compounds. As the battery is charged, this process is reversed: Oxygen (O2) is released to the air at the positive electrode, while the alkali metal is deposited at the negative electrode.
A number of fundamental problems stand in the way of practical implementation of such systems: insufficient rechargeability; numerous side reactions that limit the stability; and, in trials using lithium, clogging of the porous electrode by lithium peroxide. Sodium is much easier to obtain, and may be a better choice. Sodium–oxygen cells surprisingly do not produce sodium peroxide, instead making mainly sodium superoxide (NaO2), which can be almost reversibly converted back to the elements during charging.
The system also requires an anhydrous, aprotic solvent (that cannot release any H+ ions) for the electrolyte. Dimethylsulfoxide (DMSO) is a good choice for electrochemical applications, but it unfortunately reacts with sodium to form products that can be problematic.
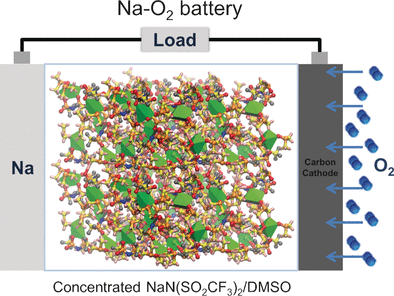
Mingfu He, Kah Chun Lau, Yiying Wu, and their team at the Ohio State University, California State University, and Argonne National Laboratory (USA) have now found an approach to solve this problem. In their system, a very high concentration of the organic salt sodium trifluoromethanesulfonimide (NaTFSI) stabilizes DMSO in the presence of sodium.
By using Raman spectroscopy of NaTFSI/DMSO electrolyte solutions in conjunction with computational simulations, the scientists were able to explain why this is so. Highly concentrated solutions result in a structure of loosely crosslinked Na(DMSO)3TFSI units that binds up a large proportion of the DMSO molecules, leaving only a few available for a reaction. The sodium then preferentially attacks the TFSI anions, which is advantageous because the product forms a passivating protective layer on the sodium electrode.
The researchers built a small battery with this system. It demonstrated good electrochemical properties and underwent 150 charge/discharge cycles without any notable loss of efficiency. In contrast, cells with a dilute electrolyte solution could only last for 6 cycles.
More information: Mingfu He et al. Concentrated Electrolyte for the Sodium-Oxygen Battery: Solvation Structure and Improved Cycle Life, Angewandte Chemie International Edition (2016). DOI: 10.1002/anie.201608607
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley