Silver nanoparticles' protein 'corona' affects their toxicity
A senior fellow at the Faculty of Chemistry, MSU, Vladimir Bochenkov, together with his colleagues from Denmark, have established the mechanism of interaction of silver nanoparticles with the cells of the immune system. The study is published in the journal Nature Communications.
"Currently, a large number of products contain silver nanoparticles—antibacterial drugs, toothpaste, polishes, paints, filters, packaging, medical and textile items. The functioning of these products lies in the capacity of silver to dissolve under oxidation and form ions Ag+ with germicidal properties. At the same time, there are in vitro research data showing silver nanoparticles' toxicity for various organs, including the liver, brain and lungs. In this regard, it is essential to study the processes occurring with silver nanoparticles in biological environments, and the factors affecting their toxicity," says Vladimir Bochenkov.
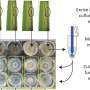
The study is devoted to the protein corona—a layer of adsorbed protein molecules that is formed on the surface of the silver nanoparticles during their contact with the biological environment, for example, in blood. This protein corona masks nanoparticles and largely determines their fate, including the speed of the elimination from the body, the ability to penetrate to a particular cell type, the distribution between the organs, etc.
According to the latest research, the protein corona consists of two layers: a rigid hard corona consisting of protein molecules tightly bound with silver nanoparticles; and a soft corona, consisting of weakly bound protein molecules in a dynamic equilibrium with the solution. Until now, the soft corona has been studied very little because of experimental difficulties—the weakly bound nanoparticles that were separated from the protein solution easily desorbed, leaving only the rigid corona on the nanoparticle surface.
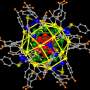
The size of the studied silver nanoparticles was 50 to 88 nm, and the diameter of the proteins that made up the crown were three to seven nm. Scientists managed to study the silver nanoparticles with the protein corona in situ, without removing them from the biological environment. Due to the localized surface plasmon resonance used for probing the environment near the surface of the silver nanoparticles, the functions of the soft corona have been primarily investigated.
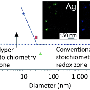
"In the work, we showed that the corona may affect the ability of the nanoparticles to dissolve to silver cations Ag+, which determine the toxic effect. In the absence of a soft corona (quickly sharing the medium protein layer with the environment), silver cations are associated with the sulfur-containing amino acids in serum medium, particularly cysteine and methionine, and precipitate as nanocrystals Ag2S in the hard corona," says Vladimir Bochenkov.
Ag2S (silver sulfide) famously easily forms on the silver surface even on the air in the presence of the hydrogen sulfide traces. Sulfur is also part of many biomolecules contained in the body, provoking the silver to react and be converted into sulfide. Formation of Ag2S nano-crystals due to low solubility reduces the bioavailability of the Ag+ ions, reducing the toxicity of silver nanoparticles to null. With a sufficient amount of the amino acid sulfur sources available for reaction, all the potentially toxic silver is converted into the nontoxic insoluble sulfide. This is what happens in the absence of a soft corona.
In the presence of a soft corona, the Ag2S silver sulfide nanocrystals are formed in smaller quantities or not formed at all. Scientists attribute this to the fact that the weakly bound protein molecules transfer the Ag+ ions from nanoparticles into the solution, thereby leaving the sulfide uncrystallized. Thus, the soft corona proteins are vehicles for the silver ions.
This effect, scientists believe, should be taken into account when analyzing the stability of silver nanoparticles in a protein environment, and in interpreting the results of the toxicity studies. Studies of the cell viability of the immune system (J774 murine line macrophages) confirmed the reduction in cell toxicity of silver nanoparticles at the sulfidation (in the absence of a soft corona).
Vladimir Bochenkov's challenge was to simulate the plasmon resonance spectra of the systems involved and to create the theoretical model that allowed quantitative determination of silver sulfide content in situ around nanoparticles, following the change in the absorption bands in the experimental spectra. Since the frequency of the plasmon resonance is sensitive to a change in dielectric constant near the nanoparticle surface, changes in the absorption spectra contain information about the amount of silver sulfide formed.
Knowledge of the mechanisms of formation and dynamics of the behavior of the protein corona, and information about its composition and structure are extremely important for understanding the toxicity and hazards of nanoparticles for the human body. Protein corona formation could be used to deliver drugs in the body, including for the treatment of cancer.
More information: Teodora Miclăuş et al, Dynamic protein coronas revealed as a modulator of silver nanoparticle sulphidation in vitro, Nature Communications (2016). DOI: 10.1038/ncomms11770
Journal information: Nature Communications
Provided by Lomonosov Moscow State University