September 5, 2013 report
Separate teams develop similar method for creating non-oxidizing silver nanoparticles
(Phys.org) —Two teams working independently of each other have developed very similar methods for creating silver nanoparticles that appear impervious to rust. One of the teams was from the U.S. and published their results in the journal Nature. The other team was from China and published their results in the journal Nature Communications.
Both teams were looking for a way to create silver nanoparticles that could be used as a replacement for the more expensive gold nanoparticles (used extensively in biomedical applications). Silver is more abundant and costs less but its sensitivity to oxidation makes it unstable, and thus unsuitable for most of the applications that gold nanoparticles are used for. To use silver as a basis for such nanoparticles the research teams had to find a way to cause them to be impervious to oxidation, and that's just what both have achieved.
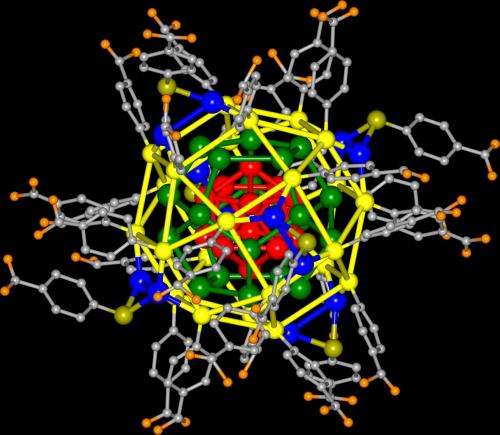
Both teams wound up with nanoparticles that were made up of exactly 44 silver atoms. Likewise both teams used a similar technique to create the nanoparticles—mixing silver nitrates with reagents in ethanol and water. The difference between the two techniques developed by the two teams was in the sulfur bearing molecules used. In either case, both resulted in the formation of a solid core of 12 silver atoms. They were surrounded by another 20 atoms that served to hold the shape of the core. The rest of the atoms were used to create a sort of cage protecting the inner structure from outside influence. The end result was a silver nanoparticle that appears to be as impervious to oxidation as gold nanoparticles—all at a greatly reduced cost. The researchers suggest it behaves like a "superatom" because its structure prevents the loss of electrons to other chemicals. Another benefit the new technique offers is preciseness—every one of the nanoparticles produced has exactly 44 silver atoms produced in exactly the same configuration, making production easier because the process is more predictable.
-

Dish containing 140 grams of silver nanoparticle powder pictured with a solution of the nanoparticles and 2 troy ounces of silver bullion, which is approximately the quantity of silver contained in the pile. Credit: Daniel Miller -

Dish containing 140 grams of silver nanoparticle powder pictured with a solution of the nanoparticles and 4 dice, which represent the shapes found within the highly symmetric cluster. They are 4 of the five Platonic solids: icosahedron, dodecahedron, octahedron, and cube. Credit: Daniel Miller
Gold nanoparticles have been used to help deliver drugs to internal bio-systems, to assist with imaging individual cells, and as biomarkers to highlight disease components. Now, it appears likely that the new technique developed by the two teams will allow for much cheaper silver nanoparticles to be used for such applications instead.
More information: 1. Ultrastable silver nanoparticles, Nature (2013) DOI: 10.1038/nature12523
Abstract
Noble-metal nanoparticles have had a substantial impact across a diverse range of fields, including catalysis, sensing, photochemistry, optoelectronics, energy conversion6 and medicine. Although silver has very desirable physical properties, good relative abundance and low cost, gold nanoparticles have been widely favoured owing to their proved stability and ease of use. Unlike gold, silver is notorious for its susceptibility to oxidation (tarnishing), which has limited the development of important silver-based nanomaterials. Despite two decades of synthetic efforts, silver nanoparticles that are inert or have long-term stability remain unrealized. Here we report a simple synthetic protocol for producing ultrastable silver nanoparticles, yielding a single-sized molecular product in very large quantities with quantitative yield and without the need for size sorting. The stability, purity and yield are substantially better than those for other metal nanoparticles, including gold, owing to an effective stabilization mechanism. The particular size and stoichiometry of the product were found to be insensitive to variations in synthesis parameters. The chemical stability and structural, electronic and optical properties can be understood using first-principles electronic structure theory based on an experimental single-crystal X-ray structure. Although several structures have been determined for protected gold nanoclusters, none has been reported so far for silver nanoparticles. The total structure of a thiolate-protected silver nanocluster reported here uncovers the unique structure of the silver thiolate protecting layer, consisting of Ag2S5 capping structures. The outstanding stability of the nanoparticle is attributed to a closed-shell 18-electron configuration with a large energy gap between the highest occupied molecular orbital and the lowest unoccupied molecular orbital, an ultrastable 32-silver-atom excavated-dodecahedral13 core consisting of a hollow 12-silver-atom icosahedron encapsulated by a 20-silver-atom dodecahedron, and the choice of protective coordinating ligands. The straightforward synthesis of large quantities of pure molecular product promises to make this class of materials widely available for further research and technology development.
2. All-thiol-stabilized Ag44 and Au12Ag32 nanoparticles with single-crystal structures, Nature Communications, DOI: 10.1038/ncomms3422
Abstract
Noble metal nanoparticles stabilized by organic ligands are important for applications in assembly, site-specific bioconjugate labelling and sensing, drug delivery and medical therapy, molecular recognition and molecular electronics, and catalysis. Here we report crystal structures and theoretical analysis of three Ag44(SR)30 and three Au12Ag32(SR)30 intermetallic nanoclusters stabilized with fluorinated arylthiols (SR¼SPhF, SPhF2 or SPhCF3). The nanocluster forms a Keplerate solid of concentric icosahedral and dodecahedral atom shells, protected by six Ag2(SR)5 units. Positive counterions in the crystal indicate a high negative charge of 4 per nanoparticle, and density functional theory calculations explain the stability as an 18-electron superatom shell closure in the metal core. Highly featured optical absorption spectra in the ultraviolet–visible region are analysed using time-dependent density functional perturbation theory. This work forms a basis for further understanding, engineering and controlling of stability as well as electronic and optical properties of these novel nanomaterials.
Journal information: Nature , Nature Communications
© 2013 Phys.org