New catalyst for hydrogen production

With the aid of platinum catalysts, it is possible to efficiently produce hydrogen. However, this metal is rare and expensive. Researchers have discovered an alternative that is just as good, but less costly.
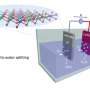
The mineral pentlandite is a potential new catalyst for hydrogen production. As described in the journal Nature Communications, it works just as efficient as the platinum electrodes commonly used today. In contrast to platinum, pentlandite is affordable and found frequently on Earth.
A team headed by Dr. Ulf-Peter Apfel and Prof. Dr. Wolfgang Schuhmann of the Ruhr-Universität Bochum describes the results together with colleagues from the Max-Planck-Institute for Coal Research in Mülheim an der Ruhr and the Technical University of Bratislava.
Producing hydrogen without precious metals
In addition to platinum, there are numerous other substances that can catalyse the reaction of water to hydrogen and oxygen and do not contain any precious metals. Among such compounds are the so-called metal chalcogenides. Usually, however, these non-metallic materials are distinctly poorer conductors of electrons and are thus inefficient catalysts.
Pentlandite consists of iron, nickel, and sulfur. Its structure is similar to the active center of hydrogenases, which are hydrogen-producing enzymes, as found, for example, in green algae. In the current study, the researchers compared the hydrogen production rate of naturally obtained and artificially produced pentlandite with platinum and other non-metallic catalysts.
Mineral pentlandite just as good as platinum
Artificial pentlandite and platinum prove to be equally good catalysts, with a performance that surpasses that of all the other materials tested. The mineral synthesized in the lab produced hydrogen much more efficiently than the naturally found variant. The reason: Inclusions of magnesium and silicon in natural pentlandite reduce its conductivity. The scientists called the output of artificial pentlandite "surprisingly high," and the rate of synthesis also remained stable for a long time.
The mineral has another advantage compared to other non-precious-metal materials. It has a greater active surface area to which the reacting substances can dock. In other non-precious-metal materials, this surface has to be created using complex methods by applying the catalyst to an electrode in the form of nanoparticles.
More information: Bharathi Konkena et al. Pentlandite rocks as sustainable and stable efficient electrocatalysts for hydrogen generation, Nature Communications (2016). DOI: 10.1038/NCOMMS12269
Journal information: Nature Communications
Provided by Ruhr-Universitaet-Bochum