How fish can metabolize toxic substances in seawater
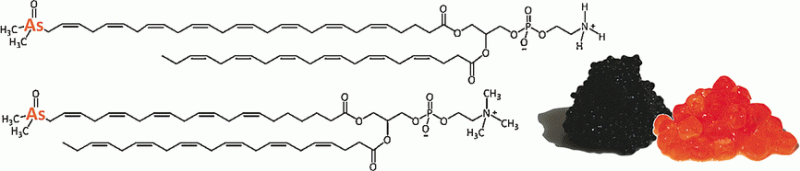
Seafish are known for their capability to accumulate toxic trace elements like mercury or arsenic. These elements are metabolized and end up chemically bound to organic compounds found in cell constituents like the membranes. Herring seems to be particularly affected: Austrian scientists have now detected a bunch of new membrane-bound arsenic substances in herring roe, as they report in the journal Angewandte Chemie.
Fish as a detoxification machine: Marine organisms have acquired various mechanisms to deal with toxic substances in seawater. Thus, seafish, prawns, and clams metabolize ingested arsenic into sugar- and lipid-bound species, which are often accumulated in the tissue. Kenneth B. Jensen, Sandra Viczek, and Kevin A. Francesconi at the University of Graz have investigated the previously unknown group of arsenolipids that had been observed at low levels in fish oil. They aimed at disclosing the relationships of these arsenolipids to phosphatidyl compounds, which are integral to membrane chemistry. Herring caviar harvested from the Norwegian Sea was chosen for the study, because the fish roe is a membrane-rich source.
The chemical species were chromatographically separated and applied to the analytical technique of mass spectrometry. In this technique, the chemical component is ionized and fragmented into smaller pieces, and the chemical composition is deduced from its fragment pattern. The scientists obtained the first results for the herring-roe samples relatively quickly. The arsenolipids could be clearly identified by their organically bound arsenic group.
The scientists found five arsenic-containing phosphatidylcholines, which are the first identified examples from this group among arsenolipids, and one member of the new group of arsenic-containing phosphatidylethanolamines. Phosphatidylcholines are a key group of chemically similar lipid compounds in the membrane. "This study identifies five of those compounds, and opens the door to discovering many more of these complex arsenic natural products", the scientists write. Why is it that so much arsenic is found in the fish membranes? "Possibly, infidelity in [the phosphatidylcholines'] biosynthesis results in the arsenic analogs," the authors write. They estimate that 50% of arsenic in herring roe can be accounted for by these compounds.
And what do these findings mean regarding toxicity of the fish caviar? Although organically bound arsenic is generally less toxic than inorganic arsenate, some arsenolipids have been proven to be highly toxic. It remains to be seen how alarming it is that fish contains more arsenic in its lipid membranes than previously known. The new arsenolipids and arsenic fatty acids are being scrutinized.
More information: Sandra A. Viczek et al. Arsenic-containing Phosphatidylcholines: a New Group of Arsenolipids Discovered in Herring Caviar, Angewandte Chemie International Edition (2016). DOI: 10.1002/anie.201512031
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley



















