Bacterial battery fuels recovery of valuable commodities from wastewater
A team at Virginia Tech has devised a system to recover ammonia from wastewater using a natural electrochemical engine: bacteria.
The discovery provides a way to remove a pollutant from waterways and capture it for commercial use.
In agriculture, ammonia is a valuable commodity. The demand for ammonia-based fertilizer is so high that the process used to produce ammonia from nitrogen gas consumes 1 percent of the world's electricity.
In wastewater, ammonia is a pollutant. If too much is discharged into a watershed, it feeds harmful algal blooms that can devastate aquatic ecosystems.
In agricultural states like Virginia, where farm runoff and animal manure already flood waterways with ammonia, and fragile ecosystems like the Chesapeake Bay, minimizing ammonia pollution in wastewater while satisfying agricultural demand answers two challenges.
"We use so much energy to convert nitrogen gas into fertilizer, and then in wastewater treatment we consume a lot of energy to convert ammonia into nitrogen gas," said Jason He, an associate professor of civil and environmental engineering in the College of Engineering. "A system that recovered the ammonia for later use, instead of just removing it, would be of great interest to sustainable wastewater treatment."
Mohan Qin, a second-year doctoral student in He's lab, has built a system that recovers ammonia and removes other contaminants while generating electricity at the same time.
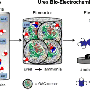
The key is combining two existing technologies: a microbial electrochemical cell, in which bacteria carry out electron-shuttling chemical reactions, and a forward osmosis cell, which uses a highly concentrated solution to draw clean water through a membrane, leaving impurities behind.
"It's a hybrid system," Qin said. "We combine the two cells to get ammonia, clean water, and electricity from wastewater."
Bacteria are naturally present in wastewater and already carry out electrochemical reactions as part of their metabolism. In an engineered system like the microbial electrochemical cell, these reactions generate electricity. The electricity drives ammonium ions present in the wastewater to the other side of the cell, creating an ammonia-rich solution.
This concentrated solution is transferred to the attached forward osmosis cell, where it draws wastewater through a membrane the same way that salt draws water out of food. Organic contaminants are left behind, and the ammonia can be easily removed and stored—leaving clean water.
The electrochemical cell also allows the electricity generated by the microbes to be harvested directly—what Qin calls a "bacterial battery." While many wastewater treatment plants produce biogas that could be converted to electricity, the costs of running and maintaining the generator are high. Qin's system bypasses the need for storing and burning biogas.
This is the first time that ammonia-driven forward osmosis has been combined with an ammonia-generating microbial electrochemical cell, Qin said. The idea earned Qin, who is from Shandong Province, China, the 2015 Innovation Award for Best Technological Advancement from the International Society for Microbial Electrochemistry and Technology.
With her bench-scale system up and running, Qin is working on computer modeling studies to explain why the system works so well, and determine what factors might improve its performance.
Meanwhile, He is pursuing opportunities to build a pilot-scale model of the combined microbial electrochemical and forward osmosis cells, working with local water treatment plants to establish a research facility.
"It's really hard to study a large-scale system in the lab," He said. "You can build it, but you can't run it: there's no wastewater. You need a place that can continuously supply wastewater."
Another microbial fuel cell system developed in He's lab is currently operating on a 200-liter scale at the Pepper's Ferry Regional Wastewater Treatment Authority in Radford, Va.
The amount of ammonia in wastewater won't eliminate the need for ammonia synthesis from nitrogen. But He and Qin's work, which is partially supported by the Institute for Critical Technology and Applied Science, can be part of a more diverse, energy-efficient approach to getting that essential nutrient—while also protecting watersheds and supplying electricity.
And as energy costs increase, using electricity to produce fertilizer from nitrogen will become less practical, and extracting energy from untapped sources—like wastewater—will become more important.
"That's the role of the researcher," He said. "To look beyond the current situation."
Provided by Virginia Tech