Study provides new insights into the genetics of drug-resistant fungal infections

A study by a multidisciplinary research team, co-directed by Worcester Polytechnic Institute (WPI), offers new insights into how virulent fungi adapt through genetic modifications to fight back against the effects of medication designed to block their spread, and how that battle leaves them temporarily weakened. These insights may provide clues to new ways to treat notoriously difficult-to-cure fungal infections like thrush and vaginitis.
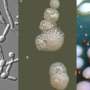
The team studied patients infected with the fungus Candida albicans (C. albicans), which causes common yeast infections and more serious bloodstream infections, who were being treated with fluconazole, one of the primary anti-fungal drugs now in use. They found that the fungus undergoes 240 genetic changes associated with drug resistance. But those changes come with a cost, they discovered. As it battles to overcome the effects of the drug, the fungus becomes weaker, with a reduction in the traits associated with virulence. The discoveries may point toward new targets for research and the potential to develop new classes of therapeutics for hard-to-treat fungal infections.
The project was co-directed by Reeta Rao, PhD, associate professor of biology and biotechnology at WPI; Dawn Thompson, PhD, and Aviv Regev, PhD, of the Broad Institute of MIT and Harvard; and Judith Berman, PhD, of Tel Aviv University. They report their findings in the paper "The evolution of drug resistance in clinical isolates of Candida albicans," published by the open-access journal eLife.
"Virtually all humans are colonized with Candida albicans, but in some individuals this benign organism becomes a serious, life-threatening pathogen," the team wrote. "Here, we used genome sequencing of isolates sampled consecutively from patients that were clinically treated with fluconazole to systematically analyze the genetic dynamics that accompany the appearance of drug resistance during oral candidiasis [infection]. Most of the genes in these clusters are not well characterized and represent new candidates involved (in) drug resistance and adaptation to the host environment."
In an accompanying eLife "Insight" piece commenting on the importance of the C. albicans drug resistance study, two researchers from the École Polytechnique Fédérale in Lausanne, Switzerland, wrote: "The work provides a global description of the genetic processes underlying drug resistance and adaptation in C. albicans. Of note, all sequencing data have been made publicly available, which is an unprecedented resource for the research community."
Thrush and vaginitis, common yeast infections caused by C. albicans, typically do not cause serious harm, but can become chronic due to a lack of drugs that can completely clear the pathogen. If a fungal infection spreads to the bloodstream (for example, via catheters or central intravenous lines), it can be deadly. Patients with compromised immune systems or implanted medical devices like pacemakers or prosthetic hips or knees, are also at greater risk for serious systemic fungal infections, which have a mortality rate between 30 and 50 percent.
In the current study, researchers sequenced the DNA and tested samples of C. albicans collected from patients with HIV who also had thrush and were being treated with fluconazole. The "azole" family of drugs do not kill the fungi—they limit growth by disrupting a protein in the yeasts' outer membrane. In many cases, some C. albicans organisms overcome the effects of fluconazole and continue to cause an infection.
Using next-generation DNA sequencing technology, the team looked for changes at the genetic level in 43 samples of C. albicans collected from the 11 patients over ten months. By sampling fungi from the same patients over time, researchers identified genetic mutations that correlated with the fungi's evolving ability to overcome the effects of fluconazole in those patients. The results revealed changes in genes associated with the structure of fungi's outer membrane and the activity of molecular pumps that can eject the drug from the yeast's cells. Numerous other genetic mutations were found to be prevalent as drug resistance increased, though the functional impact of those changes is not known.
The DNA sequencing and genomic analysis was done at the Broad Institute and Tel Aviv University. At WPI, Rao's lab tested the fitness and virulence of the C. albicans strains in the 43 samples. Those experiments found an inverse relationship between increased drug resistance and the ability of C. albicans to survive or cause additional infection. "At first that may seem counterintuitive, but it's actually a logical finding," Rao said. "It shows there is a fitness cost involved in overcoming the effect of the drug. C. albicans devotes more energy to battling the drug, so it becomes less fit and unable to cause infection until it has figured out how to overcome the effect of the drug."
Studying that window of vulnerability, when the fungus is weaker and less likely to cause more infection but not fully resistance to the drug, becomes an interesting opportunity to explore, Rao noted. Also, much more work is needed to understand the specific impact of the 240 genetic mutations found to be associated with drug resistance. Furthermore, understanding how C. albicans evades fluconazole may shed light on how some cancer cells develop resistance to current therapies because "the evolution of drug resistance in C. albicans has many parallels with the somatic evolution of cancer cells undergoing chemotherapy or treated with specific inhibitors," the authors wrote.
Journal information: eLife
Provided by Worcester Polytechnic Institute