Scientists are developing new sample holders for tiny microcrystals

Researchers of DESY, the Paul Scherrer Institute (PSI) in Switzerland and the Diamond Light Source in Great Britain have developed a new type of sample holder for so-called serial protein crystallography. The holder consists of a single crystal of silicon with a regular array of pores and allows tiny crystals to be positioned with great precision, while at the same time producing virtually no disruptive signal of its own during X-ray analysis, something that is unavoidable using conventional methods. The researchers are presenting their development in the latest edition of the journal Scientific Reports.
Determining the structure of protein structures is essential in order to understand a wealth of biological processes and therefore fundamental to developing new types of drugs. Using synchrotron radiation, such protein structures can be resolved down to an atomic level when they are examined in a crystalline form. However, many proteins are extremely difficult, sometimes even impossible, to align in the form of larger crystals. Recent developments in X-ray sources with increasing brilliance, such as DESY's PETRA III or the new generation of X-ray lasers, have opened the door to examining ever smaller crystals, just a few micrometres across, which have until now left a non-sufficient signal in crystallographic measurements. These microcrystals are considerably easier to "grow", but because of their small size they call for new approaches in preparing specimens and an improvement in the signal-to-noise ratio, because the scattered signal is considerably smaller than that of larger specimens.
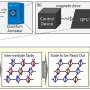
In collaboration with researchers at Diamond and PSI, scientists at DESY have now developed a new type of sample holder in which several thousand microcrystals can be positioned on a single silicon chip at the same time and then be examined by crystallographic methods. The chip can be used on the microfocus beamlines of synchrotron radiation sources, as well as with X-ray lasers such as the LCLS in Stanford and the forthcoming European XFEL. In contrast to the methods typically used so far, such as liquid jets or lipidic cubic phase (LCP) jets, in which the microcrystals are surrounded by a liquid or a gel and then analysed using X-rays, in the new sample holder the crystals are held in small holes in a membrane made from a single silicon crystal, just ten micrometres thick, which can then be scanned by an X-ray beam. This technique not only ensures that the crystals can be accurately located; the ratio of the specimen to the carrier inside the X-ray beam is also much higher, and therefore allows even smaller crystals to be examined.
"You can think of the chip as being like a sieve. The silicon membrane consists of a matrix of many tiny holes which are slightly smaller than the crystals themselves," explains Philip Roedig, a scientist at DESY and the principal author of the study. "In order to prepare the specimen, a drop of the mother solution containing the microcrystals is placed on top of the chip, and then the solution is drawn off from below. The crystals are left sticking in the holes, like in a sieve, and can be scanned by the X-ray beam, crystal by crystal." As a result, the number of crystals used is markedly lower than in liquid jet systems, for example, in which a large proportion of the crystals are carried past the beam without actually being hit by the X-ray pulses. The new chips theoretically give you a hit rate of 100 percent, meaning that every crystal in the array actually contributes towards determining the crystal structure. The efficient removal of the mother solution in which the crystals are normally suspended, leads to a reduction in the background signal and therefore improves the signal quality. To prevent the crystals on the chip from drying out, the chip is held in a stream of moist air during the preparation. Once the crystals are in place, the chip is immediately frozen in liquid nitrogen and then placed in the appropriate beamline.

In tests, the researchers have managed to collect high-resolution diffraction data from protein microcrystals that were smaller than four micrometres. These measurements were carried out on viral (polyhedrin) and lysozyme crystals on the microfocus beamline I24 of the Diamond Light Source in Great Britain. The small X-ray beam, measuring 7 x 7 µm2, only irradiated a single microcrystal at a time. The data collected from several hundred crystals were then merged to produce one complete set, and analysed. This allowed the researchers to obtain 3D information on the electron density distribution within the crystals with a resolution of 1.5 angstrom, or 0.15 nanometres.
Additional experiments are due to take place at the X-ray laser LCLS in Stanford in July, in which the structure of the crystals on the chip will be determined using high-intensity and ultrashort X-ray laser pulses. The intensity of the X-ray pulses will be so high that each crystal will immediately be destroyed by a single X-ray pulse, and the chip will have to be moved forward in step with the rate at which the X-ray laser emits the pulses. Given a frequency of 120 Hertz, this means that an entire chip, holding up to 20 000 microcrystals can be scanned in less than three minutes.
"Our new sample holder allows us to characterise tiny microcrystals with a unique level of efficiency," explains Alke Meents, the scientist at DESY who is in charge of the work. "This makes it not just a unique tool for X-ray lasers, but also ideally suited for experiments using polychromatic X-rays, which we could produce along a beamline in the extensions of PETRA III. By using polychromatic radiation from high-brilliance radiation sources, such as PETRA III, it will be possible to carry out measurements at speeds similar to those of X-ray lasers, while reducing the number of microcrystals that are needed."
More information: "A micro-patterned silicon chip as sample holder for macromolecular crystallography experiments with minimal background scattering." Sci. Rep. 5, 10451; DOI: 10.1038/srep10451
Journal information: Scientific Reports
Provided by Helmholtz Association of German Research Centres