Scientists investigate tiny protein crystals with synchrotron radiation
At DESY, scientists from the Center for Free-Electron Laser Science (CFEL), the European Molecular Biology Laboratory (EMBL) and the universities of Hamburg and Lübeck created new analysis possibilities for delicate biomolecules. At the X-ray source PETRA III, the scientists X-rayed micrometre-sized crystals of a key enzyme involved in sleeping sickness, thereby determining its atomic structure. The obtained structural data correspond to earlier analyses of the same enzyme at the world's most powerful X-ray laser in Stanford (US). "Since only a few of these valuable crystals are needed, this method opens up new possibilities for the structural analysis of biomolecules," said CFEL scientist Cornelius Gati, lead author of a research study which describes this new method in IUCrJ, the high-profile scientific journal of the International Union of Crystallography.
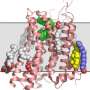
From the structure of a biomolecule at the atomic-level, biologists can determine its functioning and properties, thus for example allowing the design of tailor-made medication. For decoding the highly complex molecules, scientists use X-ray crystallography. By placing a biomolecular crystal in an X-ray beam, they can measure the pattern of X-ray light that encodes the three-dimensional structure of the constituent molecules of the crystal. The molecular structure is decoded using sophisticated computational methods. "The principle can be explained as a kind of mathematical microscope," said Gati to illustrate this method established about a hundred years ago by the German X-ray pioneer Max von Laue and by the British scientists William Henry Bragg and William Lawrence Bragg, and which today is routinely used to investigate matter in many fields.
In contrast to other materials, many biomolecules are difficult to crystallise. "It requires a considerable effort to grow large enough crystals from complicated proteins for standard analyses," said Lars Redecke from the joint Laboratory for Structural Biology of Infection and Inflammation of the universities of Hamburg and Lübeck. Some biomolecules at best form microcrystals which until now could only be analysed with the extremely intense light of X-ray lasers. These X-ray lasers are large-scale facilities. So far there are only two operating X-ray FELs in the world. The world's most modern X-ray laser, European XFEL, is currently being built, running from the DESY campus in Hamburg to the neighbouring town of Schenefeld in Schleswig-Holstein.
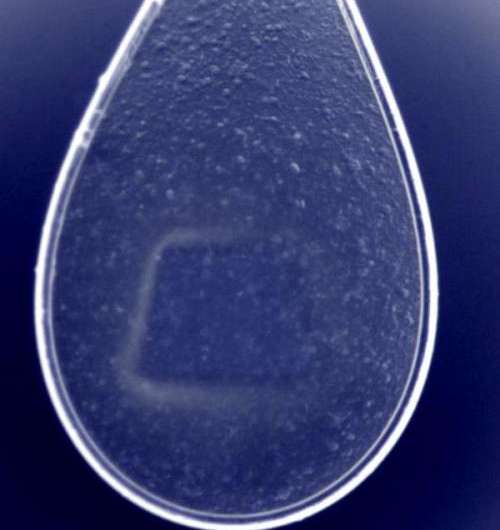
At DESY's X-ray source PETRA III, the team has now found a way to investigate microcrystals with more common research light sources as well – with the so-called synchrotron radiation sources. They grew microcrystals from the enzyme cathepsin B of the Trypanosoma brucei parasite, which causes sleeping sickness. These needle-shaped crystals typically had a length of 4 to 15 thousandths of a millimetre (micrometre). Like making soap-bubbles, the scientists use a small nylon loop to extract a 20-micrometre thin film from an aqueous crystal suspension, with about 5000 microcrystals in each film.
The prepared nylon loop was then mounted into the measuring equipment and scanned with the fine and intense X-ray beam of DESY's synchrotron radiation source PETRA III. The beam at the measuring station P14, operated by the European Molecular Biology Laboratory EMBL, has a diameter of barely five thousandths of a millimetre and it gradually scanned all crystals in the loop, with each part of the sample being exposed about half a second before the intense X-rays destroyed the investigated crystal. With this method, the scientists were able to determine from 80 crystals the atomic structure of the enzyme with a spatial resolution of 0.3 millionth of a millimetre (nanometre). "With a volume of 10 cubic micrometres, these crystals are among the smallest protein crystals that have been analysed with synchrotron radiation," EMBL scientist Thomas Schneider points out.
"Our method is especially interesting for difficult to crystallise molecules," said Redecke. "An advantage of this procedure is that we can work with components that are existing or will be available in the future at many research light sources," explains Gleb Bourenkov from EMBL, the second lead author of the study. However, not all synchrotron radiation sources can perform this analysis. It requires a very fine X-ray beam of especially high intensity – both provided at PETRA III, which today is the world's most brilliant synchrotron X-ray light source.
The new method also optimises the utilisation of the bright X-ray light. Whereas for standard analyses of larger crystals the bright X-ray light has to be dimmed to avoid rapid damage of the delicate biomolecules, the new procedure exploits the maximum power of PETRA III for biomolecule crystallography for the first time. "This approach will complement X-ray laser studies of difficult biological samples, allowing researchers to make optimal use of facilities that are in high demand," said CFEL scientist Henry Chapman. Since this technology is also valuable for other research light sources, several research centres already expressed their interest.
More information: "Serial crystallography on in vivo grown microcrystals using synchrotron radiation"; Cornelius Gati, Gleb Bourenkov, Marco Klinge, Dirk Rehders, Francesco Stellato, Dominik Oberthür, Oleksandr Yefanov, Benjamin P. Sommer, Stefan Mogk, Michael Duszenko, Christian Betzel, Thomas R. Schneider, Henry N.Chapmana, and Lars Redecke; IUCrJ (February 2014); DOI: 10.1107/S2052252513033939
Provided by Deutsches Elektronen-Synchrotron