Incorporation of DOPA into engineered mussel glue proteins
Mussels can do it, but we haven't been able to: gluing under water. In the journal Angewandte Chemie, a team consisting of Korean, Indian, and Canadian scientists has now introduced a new method that makes it possible to synthesize sticky mussel proteins. The key to their success is the "deception" of a bacterial enzyme that makes it possible to incorporate the amino acid DOPA, which is crucial for the adhesive effect.
Gluing wet surfaces or even under water remains a major challenge. Mussels, on the other hand, have it worked out: metal ships' hulls, wooden posts, stones, or plastic buoys—mussels stick fast to practically anything. It would be very useful if we could do that too, so we could easily seal leaks in underwater pipelines and close bleeding wounds during operations.
Unfortunately, it isn't possible to isolate enough of this all-purpose, underwater glue from mussels. Biotechnological production of the adhesive mussel proteins in bacterial cultures has also been unsuccessful. The main problem has been the inability of researchers to incorporate an amino acid that is present in large quantities in the mussel glue and is critical to its adhesive action. This amino acid, dihydroxyphenylalanine (DOPA) is a modified form of the amino acid tyrosine. Mussel can modify tyrosine groups in adhesive proteins to DOPA—bacteria are not able to perform this modification.
Yet, DOPA plays a key role by crosslinking the glue, binding to inorganic oxides in stone, and forms hydrogen bridge bonds to diverse materials. By binding multivalent metal ions like iron from seawater, it gives the mussel glue self-healing properties.
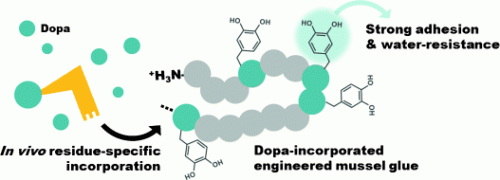
Researchers at Pohang University of Science and Technology (POSTECH) and Chungnam National University in Korea, as well as Central Leather Research Institute in India and University of Alberta in Canada have now been able to induce bacterial cultures to incorporate DOPA by "outwitting" an enzyme.
The team headed by Hyung Joon Cha changed the way to incorporate DOPA: instead of modifying tyrosine to DOPA after protein synthesis, incorporation of DOPA itself during protein synthesis was tried. During protein synthesis, amino-acid-specific enzymes load amino acid specific transfer molecules (tRNAs) with the corresponding amino acid. The tRNA recognizes the genetic code for its amino acid and delivers it so that it can be incorporated into the growing protein chain. Fortunately, the bacterial transfer specialist for tyrosine is relatively loose: If no tyrosine is present, it accepts DOPA as well. The scientists thus remove the tyrosine from the culture medium and add DOPA instead. This resulted in a protein with a high DOPA concentration that adheres to a variety of dry and wet surfaces much like the natural mussel glue.
More information: Yang, B., Ayyadurai, N., Yun, H., Choi, Y. S., Hwang, B. H., Huang, J., Lu, Q., Zeng, H. and Cha, H. J. (2014), "In Vivo Residue-Specific Dopa-Incorporated Engineered Mussel Bioglue with Enhanced Adhesion and Water Resistance." Angew. Chem. Int. Ed.. doi: 10.1002/anie.201406099
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley