Minimizing the cellular contaminants that plague the differentiation of pluripotent stem cells
Embryonic stem cells (ESCs) have the potential to form more than 200 distinct cell types in the human body. Although ESCs can differentiate into any specialized tissue, scientists are still unsure of how to coax these pluripotent cells to reliably form a desired cell type without producing a mix of contaminating cell lineages.
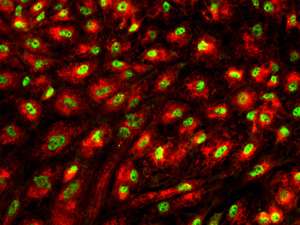
A research team led by Bing Lim from the A*STAR Genome Institute of Singapore has now developed a new method for directing ESCs into highly pure populations of liver cells or pancreas cells. These untainted populations of organ-specific cells could form the basis of future therapies or be used as platforms upon which to screen drugs.
"Heterogeneous mixtures of cell types are unsuitable for transplantation or other therapeutic purposes," says Kyle Loh, a previous member of Lim's lab now based at the Stanford University School of Medicine in the United States. "We can precisely differentiate embryonic stem cells into a pure population of a given lineage of interest," he explains.
Lay Teng Ang of the liver research program in Lim's lab sought a way to guide human ESCs to differentiate reliably into endoderm—the cell type that gives rise to organs including the lungs, liver and intestines. To do this, Ang systematically perturbed developmental signals at four consecutive steps of endoderm formation, searching for molecules that could produce a single, desired cell type as well as block the induction of unwanted alternatives. Along the way, the researchers generated what Lim calls "a roadmap for endoderm differentiation."
Their strategy showed that a variety of growth factors and signaling proteins initially help transform ESCs into endoderm progenitor cells. However, the researchers had to inhibit these same molecules within 24 hours to prevent the cells from turning into another tissue type known as mesoderm.
Knowledge of this timing and the downstream signaling dynamics eventually allowed Lim's team to differentiate the ESCs into pure populations of liver and pancreas cells, while excluding other lineages at each developmental branch point. The researchers also noted the endodermal enhancers existed in a surprising diversity of 'pre-enhancer' states as uncommitted cells prior to activation: they also documented the 'permissive' chromatin marks that provide ESCs with their flexible developmental capacity.
"We first needed to understand the signals that control stem cell differentiation—and thus what controls lineage splitting," Loh says. "Then we could unilaterally repress differentiation of ESCs towards other cell types and instead drive stem cells exclusively toward a uniform population of desired stem cells."
More information: Loh, K. M., Ang, L. T., Zhang, J., Kumar, V., Ang, J. et al. "Efficient endoderm induction from human pluripotent stem cells by logically directing signals controlling lineage bifurcations." Cell Stem Cell 14, 237–252 (2014). dx.doi.org/10.1016/j.stem.2013.12.007
Journal information: Cell Stem Cell