SON ensures proper splicing of proteins to keep embryonic stem cells in a state of self-renewal
Embryonic stem cells (ESCs) retain their ability to self-renew indefinitely thanks to a number of proteins known as pluripotency factors. These factors must be modified at the RNA level before they are turned into proteins in order to do their job properly.
An international team, including researchers from A*STAR, has now found that a protein called SON acts as a master regulator of RNA splicing in these pluripotency factors and is essential to keep human ESCs in their undifferentiated state.
"This is the first study to demonstrate the importance of splicing regulation in human ESCs," says Huck-Hui Ng, executive director of the A*STAR Genome Institute of Singapore.
Ng and his co-workers previously identified SON in a genome-wide screen designed to discover proteins needed for ESCs to maintain their 'stemness'— their ability to self-renew and differentiate into diverse cell types. Out of more than 20,000 genes tested, the gene encoding SON—a protein others had previously linked with splicing functions—ranked in the top ten of potential regulators of pluripotency. Ng's team, led by Xinyi Lu and Jonathan Göke, manipulated SON expression levels in human ESCs. In response to SON depletion, the cells underwent differentiation and cell death.
The researchers used RNA sequencing to better understand how SON helps to keep ESCs in a pluripotent state. In the absence of SON, they discovered that splicing patterns were altered in more than 1,000 different RNA transcripts, including many involved in maintaining ESC pluripotency and self-renewal.
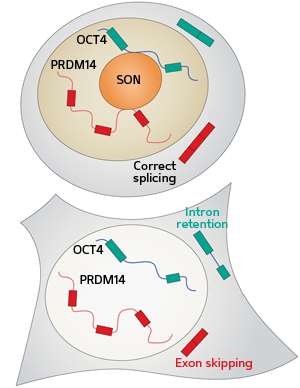
Assays designed to reveal RNA-protein interactions showed that SON bound directly to the RNA transcripts of these pluripotency-related genes. Without SON, noncoding introns were often retained and protein-coding exons often skipped. This resulted in aberrant RNA transcripts that could not maintain the stem cell properties of the ESCs.
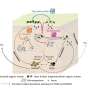
The researchers propose a model in which SON interacts with other factors in the spliceosome—the large and complex molecular machinery involved in RNA splicing—to ensure that RNA transcripts essential for ESC identity are packaged correctly (see image).
"This study should spark interest in the role of RNA processing in the maintenance of pluripotency," says Göke. One application is in the field of cellular reprogramming. For instance, to make embryonic-like stem cells from adult tissues, scientists could manipulate SON or other RNA-binding proteins to ensure proper splicing of pluripotency-associated genes. "It is imaginable," says Göke, "that regulation of RNA processing pathways can be tuned to improve the induction of pluripotency."
More information: Lu, X., Göke, J., Sachs, F., Jacques, P.-É., Liang, H. et al. SON connects the splicing-regulatory network with pluripotency in human embryonic stem cells. Nature Cell Biology 15, 1141–1152 (2013). dx.doi.org/10.1038/ncb2839
Chia, N.-Y., Chan, Y.-S., Feng, B., Lu, X., Orlov, Y. L. et al. A genome-wide RNAi screen reveals determinants of human embryonic stem cell identity. Nature 468, 316–320 (2010). dx.doi.org/10.1038/nature09531
Journal information: Nature Cell Biology , Nature