March 24, 2014 feature
Nanostructures enhance light trapping for solar fuel generation
(Phys.org) —As the world's dependence on fossil fuels causes ever-increasing problems, researchers are investigating solar fuels as an alternative energy source. To make solar fuels, sunlight is converted into hydrogen or another type of chemical energy. Compared to energy produced by solar cells, which convert sunlight directly to electricity, solar fuels such as hydrogen have the advantage of being easier to store for later use.
Because of the enormous amount of sunlight that reaches Earth, solar fuel generation has the potential to serve as a clean, terawatt-scale global energy source. But in order for this to happen, the photocatalysts that enhance light absorption and light trapping must be improved, both in terms of higher performance and lower cost.
In a new study, researchers Soo Jin Kim, et al., at the Geballe Laboratory for Advanced Materials in Stanford, California, have demonstrated that photocatalysts made from iron oxide exhibit substantial performance improvements when they are patterned with nanostructures. Their paper is published in a recent issue of Nano Letters.
"I think the most significant advance is that the work will provide valuable guidelines for the design of new, nanostructured photocatalyst materials capable of effectively absorbing light and driving catalytic reactions," Professor Mark L. Brongersma at Stanford told Phys.org. "Hopefully, it will stimulate more research on photon management for photocatalyst materials. The use of photon managements in solar fuel generation is lagging behind strongly with respect to development of photon management strategies for solar cells."
As the researchers explain, iron oxide in the hematite phase (Fe2O3) is an earth-abundant semiconductor with a bandgap energy of 590 nm, which is considered close to optimal for water splitting and hydrogen production. Because it absorbs photons across a relatively large portion of the solar spectrum, it outperforms other catalyst materials that absorb smaller portions of the solar spectrum.
Despite these advantages, hematite has a weakness: it cannot absorb photons near its surface, which results in many of the photoexcited carriers recombining rather than participating in chemical reactions to produce hydrogen. This problem occurs due to a mismatch between hematite's very short (nanometer scale) carrier diffusion length compared to the absorption depth of light (micrometer scale near the surface). So even though the photons are present, they cannot be effectively used.
Previous research has attempted to address this problem by adding metal nanostructures to enhance light absorption in the near-surface region of the photocatalysts. However, this approach suffers from intrinsic optical losses in the metal.
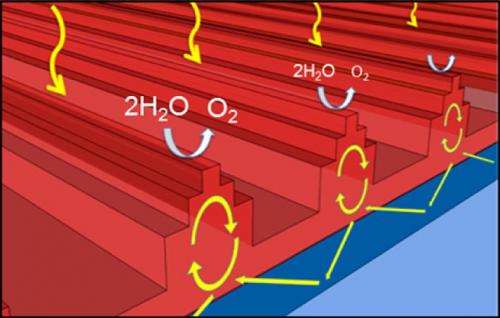
In the current study, the researchers have circumvented this problem of optical loss by nanopatterning the hematite photocatalysts themselves. The nanostructures allow the photocatalyst to overcome the detrimental mismatch between the carrier diffusion and photonic absorption length scales, and redistribute the photons to the near-surface region.
Nanostructuring's benefits come from the fact that it allows sunlight to drive optical resonances in the hematite, resulting in an enhancement of both light absorption and light scattering. By engineering the size, shape, spacing, and dielectric environment of the nanostructures, the researchers could optimize and tune the resonant wavelengths across the solar spectrum.
This strategy of nanostructuring a photocatalyst could be extended to other photocatalyst materials. As nanopatterning techniques continue to become used more often in many different areas, it is likely that nanostructured arrays can be made inexpensively over large areas.
"Next, we are going to employ metamaterials concepts in our photocatalyst materials," Brongersma said. "We will see where it takes us!"
More information: Soo Jin Kim, et al. "Light Trapping for Solar Fuel Generation with Mie Resonances." Nano Letters. DOI: 10.1021/nl404575e
Journal information: Nano Letters
© 2014 Phys.org. All rights reserved.