Genetic engineering: Workhorse bacteria deliver in an acidic environment
A technique that simultaneously modifies multiple genes in fermentation bacteria could aid lactic acid production for industry.
Many industries harness the prowess of bacteria that produce lactic acid by fermenting sugar. The bacteria, typically Lactobacillus pentosus, produce the acid in specialized tanks at an optimal pH range of 5.0–5.5. To maintain this range, neutralizing agents such as calcium hydroxide must be added to the fermentation vessel. Adding these agents, however, can lead to the formation of unwanted salts and complicate downstream processing.
Jin Chuan Wu at the A*STAR Institute of Chemical and Engineering Sciences and co‐workers in Singapore have now devised a route to more acid-tolerant lactic acid bacteria1. Wu explains that fermenting sugars at a lower pH would reduce the need for neutralizing agents, making the downstream process easier and reducing the cost of production.
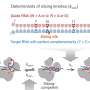
Over 70 genes are involved in the acid tolerance of bacteria. Modifying these genes sequentially would be time-consuming and complex. As a workaround, Wu and his team used a technique known as error-prone whole genome amplification to modify numerous genes at the same time. The team first reported this method in 20112.
To apply the technique, the researchers first isolated and copied the genomic DNA of L. pentosus using the polymerase chain reaction (PCR) DNA amplification technology. The intentional use of imperfect reaction conditions during this amplification process led to the desired introduction of multiple genetic copying errors. Wu and co-workers repeated this error-introducing PCR cycle 50 times, creating numerous mutants of the original DNA sequence. They then introduced the mutated DNA sequences back into the bacteria—a method known as transformation. Next, the researchers screened the different strains of mutant bacteria to assess their acid tolerance and checked their ability to ferment carbohydrates to ensure that it remained unchanged.
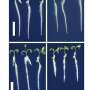
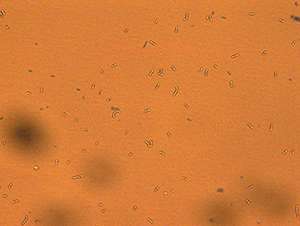
One mutant bacterial strain (see image) could tolerate a pH of 3.8 and still ferment effectively in the more acidic environment. Wu and his team found that the technique caused the genes relevant to acid tolerance to mutate positively, while the genes relevant to lactic acid production suffered no negative effects. Additionally, the team discovered that the new strain could be cultured—or grown—25 times before any changes to the desired genetic sequence occurred.
"Next, we plan to use this technique to further improve the acid tolerance of our newly isolated thermophilic lactic acid bacteria for potential industrial applications for lactic acid production," says Wu. The food, textile and cosmetic industries, for example, all use lactic acid.
The A*STAR-affiliated researchers contributing to this research are from the Institute of Chemical and Engineering Sciences
More information: Ye, L., Zhao, H., Li, Z. & Wu, J. C. Improved acid tolerance of Lactobacillus pentosus by error-prone whole genome amplification. Bioresource Technology 135, 459–463 (2013). | article: dx.doi.org/10.1016/j.biortech.2012.10.042
Luhe, A. L., Tan, L., Wu, J. & Zhao, H. Increase of ethanol tolerance of Saccharomyces cerevisiae by error-prone whole genome amplification. Biotechnology Letters 33, 1007–1011 (2011). | article: link.springer.com/article/10.1007%2Fs10529-011-0518-7
Journal information: Bioresource Technology