Metal-free catalyst outperforms platinum in fuel cell
Researchers from South Korea, Case Western Reserve University and University of North Texas have discovered an inexpensive and easily produced catalyst that performs better than platinum in oxygen-reduction reactions.
The finding, detailed in Nature's Scientific Reports online today, is a step toward eliminating what industry regards as the largest obstacle to large-scale commercialization of fuel cell technology.
Fuel cells can be more efficient than internal combustion engines, silent, and at least one type produces zero greenhouse emissions at the tail pipe. Car and bus manufacturers as well as makers of residential and small-business-sized generators have been testing and developing different forms of fuel cells for more than a decade but the high cost and insufficiencies of platinum catalysts have been the Achilles heel.
"We made metal-free catalysts using an affordable and scalable process," said Liming Dai, the Kent Hale Smith Professor of macromolecular science and engineering at Case Western Reserve and one of the report's authors. "The catalysts are more stable than platinum catalysts and tolerate carbon monoxide poisoning and methanol crossover."
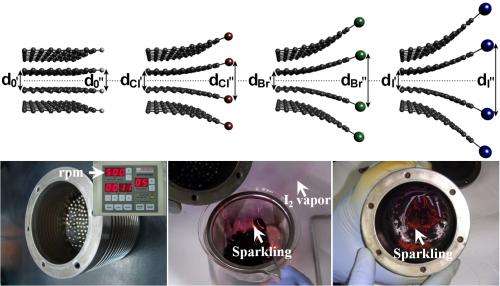
And, in their initial tests, a cathode coated with one form of catalyst—graphene nanoparticles edged with iodine—proved more efficient in the oxygen reduction reaction, generating 33 percent more current than a commercial cathode coated with platinum generated.
The research was led by Jong-Beom Baek, director of the Interdisciplinary School of Green Energy/Low-Dimensional Carbon Materials Center at South Korea's Ulsan National Institute of Science and Technology. Fellow authors include: In-Yup Jeon, Hyun-Jung Choi, Min Choi, Jeong-Min Seo, Sun-Min Jung, Min-Jung Kim and Neojung Park, from Ulsan; Sheng Zhang from Case Western Reserve; and Lipeng Zhang and Zhenhai Xia from North Texas.
Like a battery, a fuel cell converts chemical energy into electrical energy. It works by removing an electron from a fuel, usually hydrogen or methanol mixed with water, at the cell's anode, or positive electrode, creating a current.
Hydrogen ions produced then pass through a membrane to the cathode, or negative electrode. Here, oxygen molecules from the air are split and reduced by the addition of electrons and combined with the hydrogen ions to form water and heat—the only byproducts.
A better, cheaper catalyst than scarce and costly platinum is required if hydrogen fuel cells and direct methanol fuel cells are to become realistic alternatives to fossil fuels, the authors say.
The technology to make alternative catalysts builds on a simple and cheap industrial process several of the researchers developed to make graphene sheets from graphite.
Inside a ball miller, which is a canister filled with steel balls, the researchers broke graphite down into single-layer graphene nanoparticles. While the canister turned, they injected chlorine, bromine or iodine gas to produce different catalysts.
In each case, gas molecules replaced carbon atoms along the zigzag edges of nanoplatelets created by milling. Not only were the edges then favorable to binding with oxygen molecules, but the bond strength between the two oxygen atoms weakened. The weaker the oxygen bonds became, the more efficiently the oxygen was reduced and converted to water at the cathode.
In testing, a cathode coated with iodine-edged nanoplatelets performed best. A cathode coated with bromine-edged nanoparticles generated 7 percent less current than the commercial cathode coated with platinum, the chlorine-edged nanoplatelets 40 percent less.
In a test of durability, electrodes coated with the nanoplatelets maintained 85.6 percent to 87.4 percent of their initial current after 10,000 cycles while the platinum electrodes maintained only 62.5 percent.
Carbon monoxide was added to replicate the poisoning that many scientists blame for the poor performance of platinum at the cathode. The performance of the graphene-based catalysts was unaffected.
When methanol was added to replicate methanol crossover from the anode to cathode in direct methanol fuel cells, the current density of the platinum catalyst dropped sharply. Again, the graphene-based catalysts were unaffected.
"This initial research proves such catalysts work better than platinum," Baek said. "We are working now to optimize the materials."
More information: Facile, scalable synthesis of edge-halogenated graphene nanoplatelets as efficient metal-free electrocatalysts for oxygen reduction reaction, Scientific Reports, DOI: 10.1038/srep01810
Journal information: Scientific Reports
Provided by Case Western Reserve University

















