Crystallization-induced asymmetric synthesis of nonracemic platinum(IV) polysulfide tris(chelate) complexes
(Phys.org) -- Asymmetry of molecules can make the difference between a drug and a poison. It is therefore vital to control this parameter in synthesis. Polish researchers found how stirring a reaction mixture in the presence of suitable seeding crystals can strongly affect the products. They report their findings in the European Journal of Inorganic Chemistry.
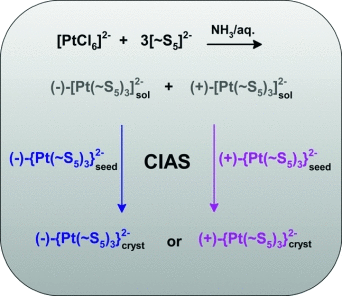
Witold Rybak and co-workers at the University of Wrocław investigated the synthesis of a platinum(IV) polysulfide anionic complex (NH4)2[Pt(S5)3]·2H2O and observed that vigorous stirring during the preparation played a unique role in determining the properties of the product obtained. Without stirring, the product was racemic, its composition did not depend upon the reactants or on the crystals used for seeding. When the reaction mixture was stirred vigorously during the whole synthesis in the presence of a particular seeding crystal, however, the properties of the product were surprisingly similar to those of the seeding crystal used. This observation and the accompanying study also led to the elucidation of the mechanism of this chiral synthesis.
Previously, the asymmetric transformation of the reaction product during crystallization was believed to determine the properties of the final product obtained; however, this assumption had not been able to explain some experimental observations. A closer study has now led to the recognition of the true mechanism: the reason behind the formation of a product of a certain configuration is a crystallization-induced autocatalytic asymmetric synthesis mechanism. The seeding crystal molecule catalyzes the formation of more molecules of its own kind.
This discovery can be used to synthesize products of the desired chirality by selecting the seeding crystal with the right configuration and performing the synthesis under continuous stirring.
More information: Witold Rybak, Crystallization-Induced Asymmetric Synthesis of Nonracemic Platinum(IV) Polysulfide Tris(chelate) Complexes, European Journal of Inorganic Chemistry , 2012, No. 23, dx.doi.org/10.1002/ejic.201200479
Journal information: European Journal of Inorganic Chemistry
Provided by Wiley