March 25, 2011 weblog
Creating power from water
(PhysOrg.com) -- Creating power from water. I bet when I say that you picture a dam or a large turbine being pushed by hundreds of thousands of gallons of water, all rushing at tremendous speeds. It is a cool, and accurate, image of how most power comes from water. That is not to say that it is the only way that power can come from water.
Researchers at the The Tata Group have been working with Daniel Nocera, an MIT scientist and founder of SunCatalytix, have found a new way to coax power out of water. If you're wondering how that was accomplished here are the basics.
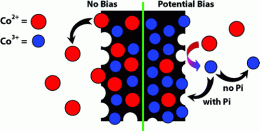
The research team placed an artificial sheet of artificial cobalt- and phosphate-coated silicon into a jar of water. This produced an effect similar to photosynthesis. The splitting of hydrogen from water was used to generate power from the sun. Interestingly enough, this technique was able to produce more power than the current generation of solar panels.
This technology, which can be used with gray water, could be used to power a mini power plant, that would be about the size of a refrigerator, according to the researchers. Of course, those plans could change, since this research is really only it its early stages. By next year the team expects to be able to power a small home with only roughly a bottle and a half of water.
While specifics of the deal have not been made public at this time, The Tata Group's mission is to being basic needs and other essential resources to low-income peoples, and the best guess is that this technology will involve that market.
More information: www.suncatalytix.com/tech.html
© 2010 PhysOrg.com



















