The sun is a star when it comes to sustainable energy
At a national scientific meeting last week where biofuels - principally ethanol - were uniformly trashed as an environmental train wreck, one bright, carbon-free light gleamed in our energy future: the sun.
Numerous sessions on energy and climate change at the annual meeting of the American Association for the Advancement in Science (AAAS), held Feb. 12-16 in downtown Chicago, were premised on the conviction that the world must drastically reduce its carbon output to stave off the worst consequences of global warming.
"The sun is absolutely a singular solution to our future energy needs," speaker Nathan Lewis, who researches synthetic photosynthesis at the California Institute of Technology, told an audience at the meeting. "Nothing else comes close. More energy from the sun hits Earth in one hour than all the energy consumed on our planet in an entire year."
While the sun's heat is already being harnessed to run steam engines - Southern California Edison last week committed itself to buying 3.4 megawatts of solar thermal power for Los Angeles customers - solar thermal and photovoltaics make up a paltry one-tenth of 1 percent of the nation's energy supply.

At a Friday the 13th AAAS session, "Basic Research for Global Energy Security: A Call to Action," Lewis, Paul Alivisatos, UC Berkeley professor of chemistry and interim director of the Department of Energy’s Lawrence Berkeley National Laboratory (LBNL), and others described new research on solar cells, batteries and transmission systems that aims either to break through current limitations of our energy technologies or to find totally new and much more efficient systems.
"This is one of the most exciting times in memory for energy research," Alivisatos said, not the least because scientists around the country anticipate increased federal funding aimed at making the country energy self-sufficient while decreasing greenhouse gas emissions.
Alivisatos, who oversees LBNL's solar energy research project, Helios, focuses his research on sunlight. His AAAS talk, "Nanoscale Materials for Solar Fuel Generation," provided examples of his laboratory's cutting edge work to improve the efficiency of solar cells and to lower their price.
Plants capture only about 1 percent of the sun's energy, which means growing plants to produce biofuels is inherently far less efficient than capturing the sun's heat with solar thermal or, even better, using photovoltaics to directly convert light to electricity. The problem with solar cells is the cost.
"We now can't afford to have it, but soon we can't afford not to have it," said Lewis at the same session. "We need to do the R&D to enable us to exploit it at scale. And to do that, we need to understand how to assemble these materials, how to exploit new physics and how to capture, convert and store the sunlight so we can bring the energy to wherever people want it when they need it."
Already, silicon-based solar cells are converting sunlight to electricity with a power efficiency of close to 25 percent, with 40 percent - and perhaps as high as 50 percent - efficiency likely with cells of layered semiconductors that optimize the absorption of red, green and blue light.
"From the point of view of physics, if you are just trying to make electricity, this problem is solved," Alivisatos said. But these efficient solar cells, made of crystalline silicon or semi-crystalline thin films of rare elements like tellurium and cadmium, are expensive to produce and, for now, affordable only in high-end applications like satellites.
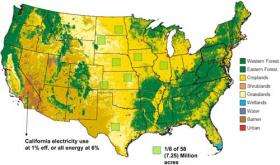
Less expensive plastic solar cells of blended polymers can now convert sunlight to energy at 5 percent efficiency, Alivisatos said. Assuming this can be improved to 8 percent efficiency, he calculates that meeting the 3.2 terawatts of demand in the United States would require covering 60 million acres with plastic solar cells. That is one-quarter of all the agricultural land in the United States, although the plan is to use marginal land, even desert, instead of cropland or grazing land.
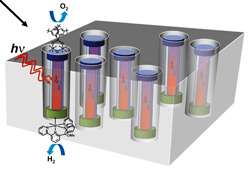
To avoid such immense solar farms, efficiencies need to go up significantly and costs need to come down. Alivisatos' goal is to produce cheap yet efficient solar cells using nanomaterials, and eventually to exploit nanomaterials as catalysts to use sunlight to split water directly into hydrogen and oxygen or to produce fuels such as methane.
Nanocrystals have a decided advantage over bulk crystalline silicon because it's easier to get perfect nanocrystals than perfect blocks of silicon, for the same reason that smaller diamonds are more common and cheaper than large ones: deep in the Earth, it's easier to create small, defect-free diamonds than large perfect stones.
Creating vast arrays of silicon-based solar cells is "like trying to coat the desert with real high quality diamonds," Alivisatos said.
Today's thin-film solar cells, which are sheets of semi-crystalline CIGS (copper indium gallium selenide) or CdTe (cadmium telluride), are similarly difficult and expensive to make.
Tiny nanocrystals, nanorods or nanowires would thus be easier and cheaper to make, but one challenge is how to pack them into a film so that the electrical charges jump readily between nanocrystals and don't get stuck. New materials such as lead selenide or a mixture of lead, selenium and sulfur work with efficiencies between 1 and 3 percent, although they can be substantially improved, Alivisatos said. Tin may work as well in place of more toxic lead.
"No nanoparticle photovoltaic has matched thin film performance yet," he said, although the electron transport through them is getting pretty good.
A new study by Alivisatos, LBNL post-doc Cyrus Wadia and UC Berkeley's Dan Kammen shows that nanocrystal forms of some abundant and inexpensive materials - iron pyrite, or fool's gold, for example - have potential as solar cell materials, with cost savings even if their efficiency is less than that of silicon.
"It turns out that some of the materials we most emphasize in our thin film industry today don't scale to enough area to solve the whole problem (of supplying the nation's electricity from sunlight)," Alivisatos said. "We need to keep our options open."
Even more ambitious, Helios's artificial photosynthesis project aims to recreate an artificial leaf that will directly convert sunlight to hydrogen fuel instead of to sugar. Alivisatos and his colleagues have already shown that nanorods of cobalt and oxygen catalyze the production of hydrogen at a level that makes it a promising catalyst for use in integrated artificial solar fuel systems. UC Berkeley chemist Peidong Yang has shown the same with titanium-coated silicon nanowires.
In his new role as interim director of LNBL, Alivisatos was emphatic that the model of the Department of Energy's Joint Bioenergy Institute, which aims to take biofuel research from lab bench to the marketplace, will work with solar energy as well.
"I think that the national labs are going to play a very important role in energy research," he said, "and I am hopeful they can act as anchor points, working with universities and companies, where this type of interaction between basic and applied research can happen in a timely way so that we can move quickly. We're in a hurry."
The annual AAAS meeting typically draws 10,000 scientists and members of the public, as well as hundreds of international and national science writers. This year, 11 UC Berkeley researchers presented papers in 12 separate sessions, on topics ranging from exoplanets (Geoffrey Marcy) and human evolution (Tim White) to the mathematics of Go (Elwyn Berlekamp).
Provided by UC Berkeley





















