October 5, 2009 feature
Scientists Use Inkjet Printer to Manipulate Genes in New Ways
(PhysOrg.com) -- With recent advances in biochemistry, researchers can control the circuitry in a developing cell, thereby influencing cells to develop into specific phenotypes. Taking a step forward in this area, researchers have recently demonstrated a new technique to control gene expression in two dimensions over time, which has not previously been demonstrated. And they have done so using a slightly modified $100 inkjet printer.
Daniel Cohen and Michel Maharbiz of the University of California, Berkeley, along with Roberto Morfino of the École Polytechnique Fédérale de Lausanne in Switzerland, have published their new method in a recent issue of PLoS ONE.
“On an engineering level, we developed an extremely low-cost, user-friendly system that any biology lab can use to explore spatiotemporal control of biological systems,” Cohen told PhysOrg.com. “On a scientific level, we demonstrated that, by controlling the position and timing of chemical dosing in a biological system, we can manipulate arbitrary genetic circuits into behaving like more complex morphogenetic circuits, which are the genetic circuits that regulate pattern formation, such as where our head is relative to our feet and why we have five fingers.”
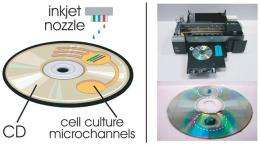
In their study, the researchers used a commercial-grade Epson R280 inkjet printer to print patterns of lactose and glucose onto an agar gel culture of E. coli in order to regulate the pathway called the lac operon. As the scientists explained, lactose acts as an inducer, while glucose acts as an inhibitor in the bistable lac operon system. The lac system has four states. It can be effectively off if there is no lactose or glucose. If there is glucose but no lactose, the system is also off. If there is glucose and lactose, the system is low-on. If there is lactose and no glucose, the system is full-on. The reason is that glucose provides more energy than lactose so it is better not to waste resources eating lactose if glucose is around.
In their new method, the researchers used a syringe pump to manually inject the lactose and glucose “inks” into the printer’s color reservoirs. The printer functions completely normally, but prints the injected solutions rather than ink. The Epson R280 also has the ability to print on rigid substrates, enabling the researchers to use normal compact discs (CDs) as substrates. Before printing, the scientists first milled 800-micron-deep wells directly into the surface of the CDs, into which they transferred the E. coli cultures. Finally, they used Adobe Photoshop to create specific patterns using the inks, and then to print them directly into the wells.
The researchers took photos of the E. coli cultures with a digital camera 15 hours after printing. They could see the patterns of lactose and glucose where they had printed in dark and light areas, respectively, due to the state of the lac operon. The bistability of the lac operon allowed for the generation of sharp gene expression boundaries, but the scientists also observed interesting dynamical behavior at boundaries between glucose and lactose areas.
Overall, using an inkjet printer to control cellular behavior could give researchers a versatile, low-cost, and open-source tool for a variety of applications in controlling gene expression.
“Fundamental science relies on the ability to manipulate specific pathways in a controlled fashion,” Cohen explained. “This allows us to learn more about the behavior of those pathways, and precise control of genetic activity is often the only way to answer questions such as what genes are responsible for what activities. On a more bio-engineering level, controlling gene expression will ultimately allow us to regulate the behavior of biological systems. Techniques such as medical gene therapy rely on this concept, while tissue engineering could greatly benefit from the ability to specify which genes are active in a given place at a given time.”
In addition, synthetic biologists are already building synthetic multicellular constructs, and could use this new method to influence the development of patterns.
“Apart from basic science, controlling genetic activity is of enormous importance in synthetic biology,” Cohen said. “For instance, people like Dr. Jay Keasling are using custom-made genetic circuits to control the production of artemisinin (an anti-malarial drug) in bacteria. Similarly, medical tissue engineering relies heavily on our ability to tell cells what to do. Sometimes the cells naturally do what we want, but other times we need to be able to specify that a stem cell differentiates into a certain lineage, or that cells in a particular area start to form blood vessels. This type of control can be achieved in many ways, one of which is through direct control of genetic activity.”
More information: Daniel J. Cohen, Roberto C. Morfino, Michel M. Maharbiz. “A Modified Consumer Inkjet for Spatiotemporal Control of Gene Expression.” PLoS ONE. September 2009, Volume 4, Issue 9, e7086. www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0007086
Copyright 2009 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.