Rust protection from nanocapsules
A particularly ingenious remedy for the problem of rust may be available soon. Scientists from the Max-Planck-Institut für Eisenforschung GmbH in Düsseldorf and the Max Planck Institute for Polymer Research in Mainz have succeeded in making two enormous strides towards developing a self-healing anticorrosion coating. In one study, they embedded a few 100-nanometre-sized polymer capsules containing anticorrosion payloads in a coating. They applied the coating to a metal and exposed the metal to corrosion through a crack in the coating. Thereupon, the capsules opened and released the protective payloads. As soon as the corrosive attack ended, the containers closed again. In the second study, the researchers encapsulated substances in nanocontainers that can heal small cracks and holes in the protective metal coating. The researchers thereby demonstrated that the containers were chemically altered and released the healing payloads when the corrosion process started. The containers then closed again at the end of the corrosive attack.
Human and animal skin is exemplary in many respects. Materials scientists are impressed above all by the way in which it heals itself when damaged. They would like to endow anticorrosion coatings with this very capacity, so that fine cracks and small holes in coatings do not spell disaster in the short or long term for the underlying metal. "We have made two breakthroughs in the quest for intelligent corrosion protection," reports Michael Rohwerder, Leader of a Research Group at the Max-Planck-Institut für Eisenforschung.
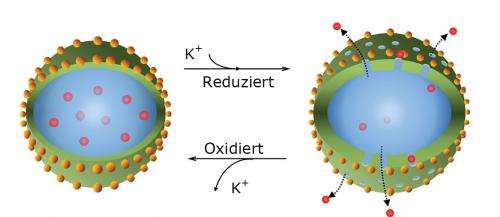
Together with their colleagues from the Max Planck Institute for Polymer Research, the Düsseldorf-based researchers tested capsules made from the conductive polymer polyaniline as containers for anticorrosive substances. They had decorated the nanocapsules with metal nanoparticles to generate suitable electrical contact between the containers and the metal to which they applied the capsules as components in a coating. Through a defect in the protective coating, they exposed the metal to corrosion by trickling a drop of salty water onto the opening in the protective coating. The corrosive attack, however, had no effect, as the walls of the polymer capsules became porous, allowing the substances contained in them to escape, which then blocked the oxygen reduction process.
The electrochemical potential is the most reliable key for opening the capsules
"What is crucial here is to select the correct signal for opening the capsule wall," says Michael Rohwerder. The capsules can thus be opened purely mechanically when the protective coating is scratched. Or they can react to a rising pH value, which can accompany the process of corrosion. However, the Max Planck team opted to exploit the electrochemical potential as a capsule opener that punctured the polyaniline cover through a process of chemical conversion. "This potential always falls when corrosion starts," explains Rohwerder. "So it provides the most reliable signal for the capsules to open." Moreover, electrical contact is required for the capsules to recognise the electrochemical alarm as well. This is provided by the metal nanoparticles between the capsule wall and the metal. The capsules detect when the corrosion has stopped through the same information channel, as the potential rises constantly at this point. The capsule wall then restructures itself and the pores are re-sealed.
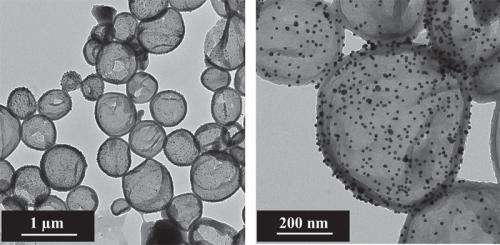
The containers, in which the researchers enclosed payloads can be used to form a polymer skin. These payloads can polymerise in a defect and seal the crack or hole. However, in this study the scientists did not apply the capsules to a metal using a coating to test them for corrosion. They replicated the chemical conditions that exist at the beginning and end of the corrosion process with reducing and oxidising substances and opened or closed the capsules in this way. "We were able to repeat this redox process with the polyaniline capsules over 80 times," says Daniel Crespy, a Research Group Leader at the Max Planck Institute for Polymer Research, who supervised the study.
Oily fluids can be encapsulated in a miniemulsion
The fact that the healing substances can be encapsulated in a targeted way is of particular interest from the chemist's perspective. This is made possible by a technique developed by researchers working with Katharina Landfester at the Max Planck Institute for Polymer Research in Mainz. They produce an emulsion from an aqueous solution, in which drops of oil float. A process that only functions to a limited extent with milk – after a while the cream accumulates on the top – was perfected by the chemists. In their miniemulsion, not only are the drops of oil similarly small in size, but they remain, thanks to a few chemical tricks, almost completely stable.
Before Daniel Crespy and his colleagues finely emulsify the oily fluid in the aqueous solution by mixing it and using ultrasound, they add the components for the polymer capsules to it. The components only react to produce long chain molecules when the chemists trickle another chemical ingredient into the prepared emulsion, which dissolves in water and triggers the polymerisation precisely on the surface of the oil drops. "This is how we can encapsulate oily fluids in an aqueous environment," says Daniel Crespy. However, despite sounding like a simple recipe, the finer details of process are actually very difficult to implement. During the polymerisation, the chemical milieu of the emulsion changes so that the drops of oil tend to aggregate and would normally accumulate on top of the water. "But we found a way of stabilising the emulsion," says Crespy.
The anticorrosion substances must be made more effective
Moreover, it is not exactly easy to prove that the capsules only release the remedy for healing the defects in a coating when necessary. To this end, the researchers in Mainz had to isolate the capsules after each step, displace them with suitable solvents and examine them with the help of nuclear magnetic resonance spectroscopy, which provided information about the volumes of the substances contained in the capsules.
In the two recent studies, the team of researchers from Düsseldorf and Mainz endowed the nanocapsules with some of the functions that a self-healing corrosion coating would have to provide. "We now want to enclose the healing substances and the anticorrosion substances together in the same capsules," says Crespy, as only both substances combined can provide comprehensive protection against the destruction caused by rust. Whereas the anticorrosion substances quickly stem the corrosion, like the initial halting of the blood flow in the case of injury, the healing substances restore the enduring anticorrosive effect of the coating. However, like a healing wound, they need more time to do their work. "Up to now, it has not been possible to encapsulate both substances under the same chemical conditions," says Daniel Crespy. This is what he and his colleagues would like to achieve.
Michael Rohwerder has also identified two further challenges that must still be overcome before the self-healing anticorrosion system is complete. "First, we must identify inhibiting substances which are as effective, for example, as chromates," says the scientist. Chromates still set the standard in terms of anticorrosion coatings at present; however, they are being banned in an increasing number of applications due to their toxicity. "Second, we must ensure that the healing substances reach a defect faster and in greater quantities," says Rohwerder. Up to now, they have been held back by the fact that they are not very water-soluble; corrosion, however, only occurs when a defect is exposed to water. If the researchers succeed in making progress on these issues, it is entirely possible that metal coatings will be the equal of living skin when it comes to powers of self-healing.
More information: Ashokanand Vimalanandan, Li-Ping Lv, The Hai Tran, Katharina Landfester, Daniel Crespy und Michael Rohwerder, Redox-Responsive Self-Healing for Corrosion Protection, Advanced Materials, online advance publication 24 September 2013; DOI: 10.1002/adma.201302989
Li-Ping Lv, Yi Zhao, Nicole Vilbrandt, Markus Gallei, Ashokanand Vimalanandan, Michael Rohwerder, Katharina Landfester und Daniel Crespy, Redox Responsive Release of Hydrophobic Self-Healing Agents from Polyaniline Capsules, Journal of the American Chemical Society, 20 August 2013; DOI: 10.1021/ja405279t
Journal information: Advanced Materials , Journal of the American Chemical Society
Provided by Max Planck Society




















