October 3, 2013 report
Researchers develop technique for enhanced reversibility of a phase-transforming material
(Phys.org) —A team of researchers at Minnesota State University has created a martensitic metal alloy that degrades very little after application of more than 16,000 thermal cycles. In their paper published in the journal Nature, the team describes how they applied mathematical theory to a zinc-gold-copper alloy and found it markedly improved the alloy's ability to withstand degradation due to multiple heating-cooling cycles.
Metals that return to a previous physical shape after being changed (bent, crumpled, etc.) using certain techniques are called a martensitic metals—typically they are created by mixing two or more metals together to create an alloy and then applying heat to cause the metal to revert back to a desired state after being deformed. Scientists have been working steadily for many years to create different metal alloys for different purposes—martensitic metals in particular are highly prized because they can be put to special purposes such as in creating stents for medical purposes. But martensitic metals have one major drawback, they can only be disfigured and reversed a few times before they start to degrade, making them unsuitable for further use. In this new effort, the research team in Minnesota has found a way to create one alloy that is able to withstand thousands of heating-cooling cycles without losing its reversibility attributes.
Their success the team explains, came about as a result of applying known mathematical theories related to stress properties of metals. Martensitic metals degrade because of changes to a transition layer between the different phases. Working the formulas, they report allowed them to eliminate this layer, effectively removing the part of the alloy that allowed for degradation.
To test their idea, the team chose a metal alloy that had previously been found to be very robust when exposed to repeated thermal cycles. They then fine-tuned the proportions of the base metal ingredients to conform with what they had worked out with the math formulas until they hit on one that was nearly impervious to repeated thermal cycles.
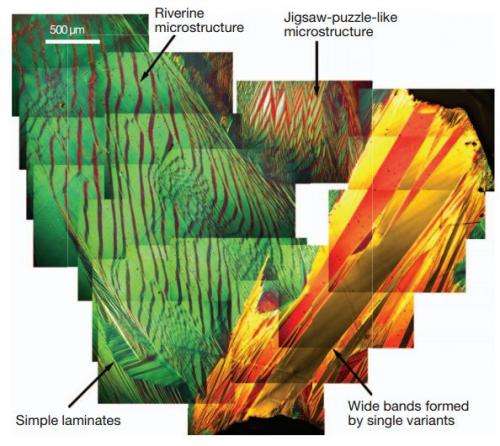
The team reports that the alloy they developed also had an unusual riverine microstructure not seen before in a martensitic metal. Perhaps more importantly, it appears that their technique might work with other alloys or non-metal materials, allowing for the creation of as yet unimagined real-world materials in a variety of applications.
More information: Enhanced reversibility and unusual microstructure of a phase-transforming material, Nature 502, 85–88 (03 October 2013) DOI: 10.1038/nature12532
Abstract
Materials undergoing reversible solid-to-solid martensitic phase transformations are desirable for applications in medical sensors and actuators, eco-friendly refrigerators and energy conversion devices4. The ability to pass back and forth through the phase transformation many times without degradation of properties (termed 'reversibility') is critical for these applications. Materials tuned to satisfy a certain geometric compatibility condition have been shown to exhibit high reversibility, measured by low hysteresis and small migration of transformation temperature under cycling. Recently, stronger compatibility conditions called the 'cofactor conditions'5, 15 have been proposed theoretically to achieve even better reversibility. Here we report the enhanced reversibility and unusual microstructure of the first martensitic material, Zn45Au30Cu25, that closely satisfies the cofactor conditions. We observe four striking properties of this material. (1) Despite a transformation strain of 8%, the transformation temperature shifts less than 0.5?°C after more than 16,000 thermal cycles. For comparison, the transformation temperature of the ubiquitous NiTi alloy shifts up to 20?°C in the first 20 cycles9, 16. (2) The hysteresis remains approximately 2?°C during this cycling. For comparison, the hysteresis of the NiTi alloy is up to 70?°C (refs 9, 12). (3) The alloy exhibits an unusual riverine microstructure of martensite not seen in other martensites. (4) Unlike that of typical polycrystal martensites, its microstructure changes drastically in consecutive transformation cycles, whereas macroscopic properties such as transformation temperature and latent heat are nearly reproducible. These results promise a concrete strategy for seeking ultra-reliable martensitic materials.
Journal information: Nature
© 2013 Phys.org


















