Researchers get a handle on what made Typhoid Mary's infectious microbes tick
Stanford University School of Medicine scientists have shown how salmonella—a bacterial menace responsible for hundreds of thousands of deaths each year from typhoid fever and food poisoning—manages to hide out in immune cells, altering their metabolism to its own benefit, much as someone might remodel a newly rented home to suit his own comfort.
Salmonella's ability to position itself inside infected people's cells for the long haul can turn them into chronic, asymptomatic carriers who, unknown to themselves or others, spread the infectious organism far and wide.
The findings, which will be published Aug. 14 in Cell Host & Microbe, could lead to new and better treatments for typhoid fever. Worldwide, there are 16 million to 20 million cases each year, resulting in about 200,000 deaths, although the disease is no longer common in developed countries. Scientists further estimate that there are close to 100 million cases of salmonella-induced gastrointestinal infections each year, the majority from food poisoning, resulting in more than 150,000 deaths.
"Between 1 and 6 percent of people infected with S. typhi, the salmonella strain that causes typhoid fever, become chronic, asymptomatic carriers," said Denise Monack, PhD, associate professor of immunology and microbiology and the study's senior author. "That is a huge threat to public health."
A classic case of a chronic, symptom-free carrier is the infamous Typhoid Mary, an Irish immigrant who made her living as a cook in and around New York City in the early 1900s. She infected about 50 people with typhoid fever. "To all outward appearances, she was perfectly healthy," Monack said.
Monack and others have sought to learn how a microbe that causes such profound acute symptoms in most people can lie dormant in symptom-free carriers' bodies for long stretches of time. To address this, Monack has developed a mouse model of persistent salmonella infection. S. typhi—the typhoid-inducing salmonella strain—infects only humans. But S. typhimurium, a closely related strain that commonly causes food poisoning in people, can infect both humans and mice. Monack's experimental mice can harbor an S. typhimurium infection for as long as two years—an entire mouse lifetime.
Previous work by Monack's team has shown that salmonella bacteria are able to take up residence inside voracious attack cells called macrophages (from the Greek words for "big eater"). These cells, which are called to the front lines of an immune assault against invading microbes, reside in tissues throughout the body and are known for their ability to engulf and digest pathogens.
Ornery critters that they are, macrophages would seem an unlikely hostel for bacteria bent on long-term survival. But, said Monack, a macrophage has two faces, depending on its biochemical environment. "Early in the course of an infection," she said, "inflammatory substances secreted by other immune cells stir macrophages into an antimicrobial frenzy. If you're not a good pathogen, you'll be wiped out after several days of causing symptoms."
But salmonella is one tough bug. And our bodies can't tolerate lots of inflammation. So, after several days of inflammatory overdrive, the immune system starts switching to the secretion of anti-inflammatory factors. This shifts macrophages into a kinder, gentler mode. Thus defanged, anti-inflammatory macrophages are more suited to peaceful activities, such as wound healing, than to devouring microbes.
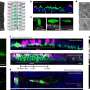
A key finding in the new study was that salmonella that survive the inflammatory assault seem to have ways of turning the subdued, anti-inflammatory macrophages into hospitable housing. Monack and her colleagues infected the experimental mice with Salmonella typhimurium and inspected their gut lymph nodes and spleen at 0, 5, 15, and 42 days post-infection for the presence of pro- and anti-inflammatory substances. They also examined the ratio of inflammatory versus anti-inflammatory macrophages, which have distinguishing surface and intracellular features.
Over time, the ratio of anti-inflammatory substances in the macrophage's surrounding environment increased, with a corresponding shift to a predominance, in the gut lymph nodes and spleen, of anti-inflammatory-macrophages. Experiments proved that at later stages of infection, S. typhimurium preferentially hung out in the anti-inflammatory macrophages. It wasn't that inflammatory macrophages were invulnerable to infection, but rather that, having infected a macrophage, S. typhimurium was much more able to replicate in the anti-inflammatory type.
After more experimentation, Monack and her colleagues came up with the probable mechanism. Salmonella apparently has a way of manipulating macrophages to expedite the switch from an inflammatory to anti-inflammatory state and, in the process, push their metabolism in a direction favorable to the bacteria.
It's known that an inflammatory macrophage gets a lot of its energy from anaerobic glucose metabolism, whereas an anti-inflammatory macrophage can get fuel from the aerobic oxidation of fatty acids, which ultimately increases the supply of glucose within the cell. A major determinant of glucose production within the macrophage is a family of intracellular receptors that, when bound by specific fatty acids from outside the cell, head to the nucleus and switch on or off numerous genes, including ones responsible for fat metabolism. These roaming genetic switches, called peroxide-proliferation-induced receptors, or PPARs, have been intensely studied. An important set of drugs—including rosiglitazone (Avandia) and pioglitazone (Actos), both licensed in the United States to treat type-2 diabetes—activate PPARs.
Other bacteria are known to hole up inside macrophages, and there have been suggestions in the literature that PPARs somehow contribute to intracellular pathogens' ability to persist in macrophages. But nobody has known why, Monack said. So, when activation levels of one of those PPAR family members, designated PPAR-delta, proved higher in her lab's S. typhimurium-infected test mice, she and her team conducted experiments to find out why, some of them involving a strain of lab mouse whose ability to produce PPAR-delta had been disabled.
The investigators found that PPAR-delta was required for salmonella to replicate inside macrophages. Even though S. typhimurium initially colonized the mice's spleen and gut lymph nodes regardless of PPAR-delta status, six weeks later it was undetectable in the PPAR-delta-deficient mice's tissues—but present to a substantial degree, as expected, in the PPAR-delta-producing mice. Additionally, the deficient mice had far fewer anti-inflammatory macrophages, and these mice's macrophages had significantly lower glucose levels.
PPAR-delta is known for sustaining fatty-acid metabolism in cells when activated, leading to increased glucose levels in cells. Intriguingly, PPAR-delta mRNA and protein levels, as well as those of a gene regulated by PPAR-delta, rose significantly in S. typhimurium-infected macrophages, suggesting that salmonella has a way of manipulating macrophages to boost PPAR-delta activity and, commensurately, glucose levels in the cell.
"Salmonella is doing something to activate PPAR-delta," Monack said. "We suspect it's releasing some as-yet-unknown PPAR-delta-stimulating virulence factor into the macrophages it infects. If we can figure out what that is, it could lead to some great anti-salmonella therapeutics with relatively fewer side effects."
Journal information: Cell Host & Microbe
Provided by Stanford University Medical Center