It's the fineness of the grind
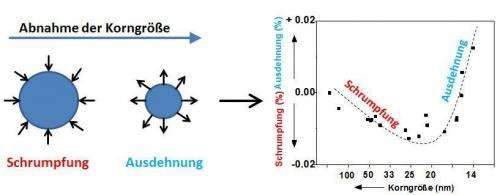
The properties of nanomaterials could be easier to predict in future. Scientists from the Max Planck Institute for Intelligent Systems in Stuttgart have ground metal into continuously finer powders in steps and prepared a detailed catalogue of how the structure of the metal grains changes depending on grain size. They discovered that the crystal lattices initially shrink, but expand again below a certain threshold grain size. The arrangement and spacing of the atoms determine numerous properties of a material. If it is possible to accurately characterise crystal lattices as a function of the particle size, it may also be possible thereby to more accurately calculate how nanoparticles of a particular size behave.
The finer coffee is ground, the more intense the taste. The relationship between properties and grind also concerns a team of scientists headed by Eric Jan Mittemeijer, Director at the Max Planck Institute for Intelligent Systems. The researchers do not work with coffee, but instead with nanoscale metals. With various very finely powdered nanograined metals they determined that the metal atoms in the individual large crystalline grains crowd together more closely, the finer the grains are ground. The crystal lattice of the material thus becomes more compressed. However, as soon as the grains measure less than approximately 30 nanometres in diameter, the atoms reverse their behaviour and the crystal lattice expands again.
Materials scientists have known for some time that the same material can exhibit various – indeed even contradictory – properties, depending on the size of its particles. This applies primarily when the dimensions of a material sample drop to the nanometre range. Quite a bit is also already known about the reasons for the different behaviour of very fine and coarser grains. In large metal crystals, most of the atoms are completely surrounded by more of the same kind of atom. In this type of ordered lattice, the attractive and repulsive forces between the metal atoms are in equilibrium.
In nanoscale grains, the surface atoms govern the material properties
By comparison, nanocrystals consist of relatively few atoms, a very large portion of which is located at the surface of the grains. As grain size falls, the ratio of surface to volume increases. The surface atoms are not surrounded on all sides by the same atoms, and below a certain crystal size they govern the material properties such as the colour, conductivity, magnetic properties and hardness of the substance.
The researchers produced nanocrystalline materials in a ball mill, pulverising nickel, iron, copper, and tungsten. The steel balls crush the metals in a cylindrically shaped drum into miniscule crystals. Through use of electron microscopy and X-ray diffraction analysis, the scientists in Stuttgart have now systematically investigated, for the first time, precisely how the atoms in finer and finer metal crystals are arranged. They were primarily interested in how the spacing between the atoms in the crystal lattice changes as a function of the size of the crystal grains.
In line with their expectations, the scientists initially observed that the crystal lattices of the four metals investigated contracted with falling grain size. "As we continued the series of experiments with smaller and smaller grains, however, we made a surprising discovery", says Eric Jan Mittemeijer. "If the granulation falls below a certain size in the nanometre range, the crystal lattice expands again and the spacing between the atoms increases".
Surface stress and excess volume compete with one another
The fact that the spacing between the atoms in the nanocrystals depends on the grain size is, according to the scientists, the result of two competing influences: surface stress and excess of free volume. In metals, the atoms, which are densely ordered in the interior and therefore possess many bonds to other atoms, have a lower energy than atoms at the surface of the grain, which are missing several bonding partners. This creates a surface stress. It permits the atoms to shift closer and closer to one another as the grain size falls and the ratio of surface to volume increases.
Below a certain size, an additional effect of the atoms at the grain's surface comes into play. A grain boundary, as it is known, forms where two nanoscale grains meet. The surface atoms of neighbouring grains, i.e. the atoms at the grain boundary, try to assume a compromise position between the two intersecting or superposed crystal lattices. They therefore displace themselves from their actual lattice locations and take up a greater volume than atoms, occupying a fixed position in a regular lattice. The researchers speak of excess free volume in the grain boundaries, which can be quite pronounced with nanomaterials. This free volume in the grain boundaries of nanomaterials creates a stress field that expands the spacing between the neighbouring atoms in the nanocrystals.
"The influence of this excess free volume on the lattice positions of the atoms can be safely neglected for objects that are larger than about 30 nanometres", says Mittemeijer. "It governs the behaviour of smaller objects, however, while the surface stress loses importance".
The research conducted by the Max Planck scientists may prove to be of great significance for materials science. "Our research contributes to better understanding the properties of nanomaterials, so that an engineer knows which nanomaterial is suitable for processing or products, for example", says Gayatri Rane, who did crucial work in the study. Sai Ramudu Meka, who also participated, adds, "If we do not know how a material behaves, we also cannot correctly employ it".
Provided by Max Planck Society