June 10, 2013 report
Polymers could help enzymes treat diseases
(Phys.org) —Conditions such as celiac disease, phenylketonuria, lactose intolerance and exocrine pancreatic disease involve abnormal enzyme activity. Enzymes administered orally could help sufferers. However, because enzymes, like all proteins, break down in the stomach and small intestine, they cannot usually survive in the gastrointestinal (GI) tract long enough to be effective. In a study published in Nature Chemistry, Jean-Christophe Leroux and his colleagues at the Swiss Federal Institute of Technology report they have found polymers that, when attached to enzymes, will prevent the enzymes from degrading in the GI tract. The research paves the way for new medical treatments.
Enzymes lose their structure and break up into their component parts easily. To correct this problem, biochemists have been attaching polymers to enzymes for decades. This makes the enzymes stable enough for use in pharmaceuticals. Doctors tend to administer these modified enzymes by injection or other non-oral routes, so they do not have to pass through the GI tract. Some oral medications contain enzymes; these have coatings to prevent stomach acid from attacking them. However, it is hard to predict how well the coatings will work in individual patients, so such medicines are not very reliable.
Recently, scientists modified an enzyme that breaks down phenylalanine, the amino acid that causes a problem for people with phenylketonuria (PKU), by combining it with the polymer polyethylene glycol (PEG). When given orally, this polymer-enzyme conjugate helped reduce phenylalanine levels in mice with PKU.
Leroux's team, which had been studying celiac disease, tried to see if it could achieve similar results by attaching polymers to enzymes that break down gluten, the protein that triggers the disease. An enzyme that digests gluten before it reaches the small intestine would benefit celiac sufferers greatly.
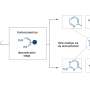
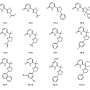
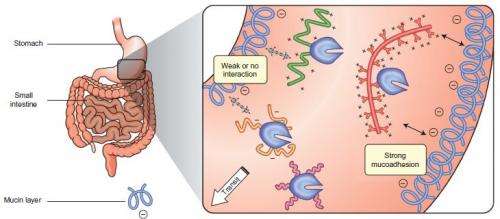
Enzymes known as proline-specific endopeptidases (PEPs) break down gluten. However, they are denaturized and deactivated in the stomach. The researchers tried to stabilize different types of PEPs by attaching different polymers to them. They labeled the modified enzymes with a fluorescent substance and fed them to rats. Enzymes attached to dendronized polymers, which have a linear main chain and dendron-type side chain, remained active in the rats' stomachs for more than three hours.
The researchers think that the dendronized polymers may provide a protective effect by adhering to the mucus in the stomach lining. They are now performing further studies on mutated enzymes to gain a better understanding of how the polymers prevent enzymes from degrading.
More information: Sustained gastrointestinal activity of dendronized polymer–enzyme conjugates, Nature Chemistry (2013) doi:10.1038/nchem.1675
Abstract
Methods to stabilize and retain enzyme activity in the gastrointestinal tract are investigated rarely because of the difficulty of protecting proteins from an environment that has evolved to promote their digestion. Preventing the degradation of enzymes under these conditions, however, is critical for the development of new protein-based oral therapies. Here we show that covalent conjugation to polymers can stabilize orally administered therapeutic enzymes at different locations in the gastrointestinal tract. Architecturally and functionally diverse polymers are used to protect enzymes sterically from inactivation and to promote interactions with mucin on the stomach wall. Using this approach the in vivo activity of enzymes can be sustained for several hours in the stomach and/or in the small intestine. These findings provide new insight and a firm basis for the development of new therapeutic and imaging strategies based on orally administered proteins using a simple and accessible technology.
Journal information: Nature Chemistry
© 2013 Phys.org