Genetically engineered spider silk for gene therapy
Genetically engineered spider silk could help overcome a major barrier to the use of gene therapy in everyday medicine, according to a new study that reported development and successful initial laboratory tests of such a material. It appears in ACS' journal Bioconjugate Chemistry.
David Kaplan and colleagues note that gene therapy — the use of beneficial genes to prevent or treat disease — requires safe and efficient carriers or "vectors." Those carriers are the counterparts to pills and capsules, transporting therapeutic genes into cells in the body. Safety and other concerns surround the experimental use of viruses to insert genes. The lack of good gene delivery systems is a main reason why there are no FDA-approved gene therapies, despite almost 1,500 clinical trials since 1989. The new study focused on one promising prospect, silk proteins, which are biocompatible and have been used in everyday medicine and medical research for decades.
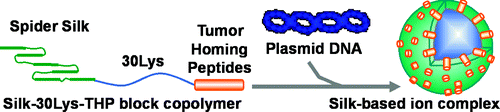
The scientists describe modifying spider silk proteins so that they attach to diseased cells and not healthy cells. They also engineered the spider silk to contain a gene that codes for the protein that makes fireflies glow in order to provide a visual signal (seen using special equipment) that the gene has reached its intended target. In lab studies using mice containing human breast cancer cells, the spider-silk proteins attached to the cancer cells and injected the DNA material into the cells without harming the mice. The results suggest that the genetically-engineered spider-silk proteins represent "a versatile and useful new platform polymer for nonviral gene delivery," the article notes.
More information: “Spider Silk-Based Gene Carriers for Tumor Cell-Specific Delivery” Bioconjugate Chem., Article ASAP. DOI: 10.1021/bc200170u
Abstract
The present study demonstrates pDNA complexes of recombinant silk proteins containing poly(l-lysine) and tumor-homing peptides (THPs), which are globular and approximately 150–250 nm in diameter, show significant enhancement of target specificity to tumor cells by additions of F3 and CGKRK THPs. We report herein the preparation and study of novel nanoscale silk-based ionic complexes containing pDNA able to home specifically to tumor cells. Particular focus was on how the THP, F3 (KDEPQRRSARLSAKPAPPKPEPKPKKAPAKK), and CGKRK, enhanced transfection specificity to tumor cells. Genetically engineered silk proteins containing both poly(l-lysine) domains to interact with pDNA and the THP to bind to specific tumor cells for target-specific pDNA delivery were prepared using Escherichia coli, followed by in vitro and in vivo transfection experiments into MDA-MB-435 melanoma cells and highly metastatic human breast tumor MDA-MB-231 cells. Non-tumorigenic MCF-10A breast epithelial cells were used as a control cell line for in vitro tumor-specific delivery studies. These results demonstrate that combination of the bioengineered silk delivery systems and THP can serve as a versatile and useful new platform for nonviral gene delivery.
Provided by American Chemical Society


















