Researchers parse the origins of hematopoietic stem cells
Researchers at the University of California, San Diego School of Medicine have identified a gene and a novel signaling pathway, both critical for making the first hematopoietic stem cells (HSCs) in developing vertebrate embryos. The discovery has implications for developing stem cell-based therapies for diseases like leukemia and congenital blood disorders.
HSCs are multipotent stem cells that give rise to all blood cell types, including red blood and immune cells. Existing medical treatments using HSCs are hampered by cell shortages and finding compatible matches between donors and recipients. Currently, it is not possible to create HSCs from converted embryonic stem cells or induced pluripotent stem cells – pluripotent cells artificially derived from non-pluripotent cells, such as skin cells.
"What we need is the ability to generate self-renewing HSCs from patients for treatments," said David Traver, PhD, an associate professor in UCSD's Department of Cellular and Molecular Medicine. "But accomplishing this goal means first understanding the mechanisms involved in creating HSCs during embryonic development."
One of those mechanisms is described for the first time in a paper published by Traver and colleagues in the June 9 issue of the journal Nature.
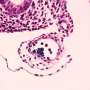
The researchers focused on a family of 19 known genes called Wnts, which had previously been recognized as important players in other aspects of embryogenesis. "Wnt signaling is involved in virtually all aspects of development," said Traver. The scientists eventually narrowed their efforts to a single, largely unstudied gene called Wnt16, which they found residing close to HSC precursors in the somites of vertebrate embryos. A somite is an early body segment in the developing embryo that gives rise to most of the adult body muscle and skeleton.
They determined that Wnt16 controls a novel genetic regulatory network necessary for HSC specification. The gene turns on key binding molecules, or ligands, that are required to definitively establish blood cell production. Knocking out Wnt16 in zebrafish, a powerful model of vertebrate development, resulted in embryos lacking HSCs.
Identifying Wnt16 as a vital factor in HSC production dramatically expands what's known about how HSCs are formed and provides new insight into what tissues talk to each other to achieve the end-goal of producing the adult blood system. "We're on the cusp of understanding something that people have been wondering about for decades," said Wilson K. Clements, first author of the study and a postdoctoral fellow in Traver's lab.
Provided by University of California - San Diego