Researchers Create Microscope With 100 Million Times Finer Resolution Than Current MRI

(PhysOrg.com) -- IBM Research scientists, in collaboration with the Center for Probing the Nanoscale at Stanford University, have demonstrated magnetic resonance imaging (MRI) with volume resolution 100 million times finer than conventional MRI.
This result, published today in the Proceedings of the National Academy of Sciences (PNAS), signals a significant step forward in tools for molecular biology and nanotechnology by offering the ability to study complex 3D structures at the nanoscale.
By extending MRI to such fine resolution, the scientists have created a microscope that, with further development, may ultimately be powerful enough to unravel the structure and interactions of proteins, paving the way for new advances in personalized healthcare and targeted medicine. This achievement stands to impact the study of materials - from proteins to integrated circuits - for which a detailed understanding of atomic structure is essential.
"This technology stands to revolutionize the way we look at viruses, bacteria, proteins, and other biological elements," said IBM Fellow Mark Dean, vice president of strategy and operations for IBM Research.
This advancement was enabled by a technique called magnetic resonance force microscopy (MRFM), which relies on detecting ultrasmall magnetic forces. In addition to its high resolution, the imaging technique has the further advantages that it is chemically specific, can "see" below surfaces and, unlike electron microscopy, is non-destructive to sensitive biological materials.
For more than a decade, IBM scientists have been making pioneering advances in
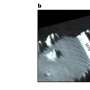
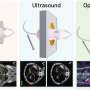
MRFM. Now, the IBM-led team has dramatically boosted the sensitivity of MRFM and combined it with an advanced 3D image reconstruction technique. This allowed them to demonstrate, for the first time, MRI on nanometer-scale biological objects. The technique was applied to a sample of tobacco mosaic virus and achieved resolution down to four nanometers. (One nanometer is one billionth of a meter; a tobacco mosaic virus is 18 nanometers across.)
"MRI is well known as a powerful tool for medical imaging, but its capability for microscopy has always been very limited," said Dan Rugar, manager of nanoscale studies, IBM Research. "Our hope is that nano MRI will eventually allow us to directly image the internal structure of individual protein molecules and molecular complexes, which is key to understanding biological function."
The new device does not work like a conventional MRI scanner, which uses gradient and imaging coils. Instead, the researchers use MRFM to detect tiny magnetic forces as the sample sits on a microscopic cantilever - essentially a tiny sliver of silicon shaped like a diving board. Laser interferometry tracks the motion of the cantilever, which vibrates slightly as magnetic spins in the hydrogen atoms of the sample interact with a nearby nanoscopic magnetic tip. The tip is scanned in three dimensions and the cantilever vibrations are analyzed to create a 3D image.
IBM Research has a distinguished history in developing microscopes for nanoscale imaging and science. IBM researchers Gerd Binnig and Heinrich Rohrer received the 1986 Nobel Prize in Physics for their invention of the scanning tunneling microscope, which can image individual atoms on electrically conducting surfaces.
Additionally, IBM has a track record dating back to the 1950s of improving healthcare through scientific achievements and collaboration with healthcare companies. In the last decade, IBM has developed a national digital mammography archive with the University of Pennsylvania; developed a clinical trial participant system with the Mayo Clinic; collaborated with Scripps to understand how influenza viruses mutate and proactively develop treatments; collaborated with European universities to develop better methods to decide on antiretroviral therapies for HIV; launched the World Community Grid, which has done projects on cancer, aids, dengue fever; and much more.
Provided by IBM