This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers unveil PI3K enzyme's dual accelerator and brake mechanisms
A group of researchers have expanded conventional knowledge on a critical enzyme that controls cell migration. In a publication in the journal Nature Communications, they reported that phosphoinositide 3-kinase (PI3K) not only acts as an accelerator to prompt cell motility, but it also has a built-in brake mechanism that impedes migration.
"PI3K is a major signaling enzyme that has been extensively studied for over 30 years due to its roles in fundamental cellular functions like growth, survival, movement and metabolism," says Hideaki Matsubayashi, lead author of the study and assistant professor at Tohoku University's Frontier Research Institute for Interdisciplinary Sciences (FRIS).
"It plays a critical part in cell migration and invasion, something that, when dysregulated, can cause many pathologies. Our work revealed that PI3K can also actively restrain these same migratory processes through a separate non-catalytic endocytic mechanism originating from its p85β subunit."
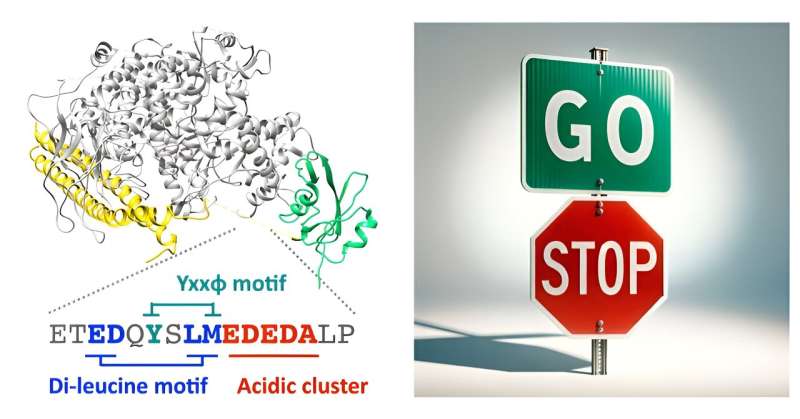
Using a combination of bioinformatics, molecular modeling, biochemical binding assays and live-cell imaging, Matsubayashi and his colleagues demonstrate that a disordered region within p85β's inter-SH2 domain directly binds to the endocytic protein AP2. This part of PI3K can activate a cellular process that pulls certain molecules into the cell, and it does so without needing the enzyme's typical lipid-modification function.
When the researchers disrupted the binding, the mutated p85β did not function as it should. Instead of regulating cell movement through its brake mechanism, it built up in specific sites within the cell. This leads to cells moving faster and more persistently, indicating a loss of the brake mechanism's control over cell migration.
"Remarkably, this single PI3K enzyme has opposing accelerator and brake pedals built into its molecular framework," adds Matsubayashi. "The endocytic mechanism helps regulate PI3K's activity to ensure that cell movement is controlled at the right times and in the right places for important biological processes."
This braking role was found to be specific to just the p85β subunit. And since the p85β subunit of PI3K is linked to cancer-promoting properties, a deeper understanding of PI3K regulation and its isoform specificity could lead to novel therapeutic strategies, such as ones that selectively inhibit the cancerous aspect of PI3K while preserving the normal functions of PI3K in healthy cells.
More information: Hideaki T. Matsubayashi et al, Non-catalytic role of phosphoinositide 3-kinase in mesenchymal cell migration through non-canonical induction of p85β/AP2-mediated endocytosis, Nature Communications (2024). DOI: 10.1038/s41467-024-46855-y
Journal information: Nature Communications
Provided by Tohoku University