This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Sustainable synthesis method reveals N-hydroxy modifications for pharmaceuticals
Recently published research results enable preparative access to novel substances that carry a modification of a structural motif frequently found in pharmaceuticals.
Sustainable chemical transformations are becoming increasingly important and in demand worldwide in all areas of life. Electrochemistry plays a decisive role in this development as electro-organic synthesis is a method that avoids large amounts of waste generated by conventional chemical transformations.
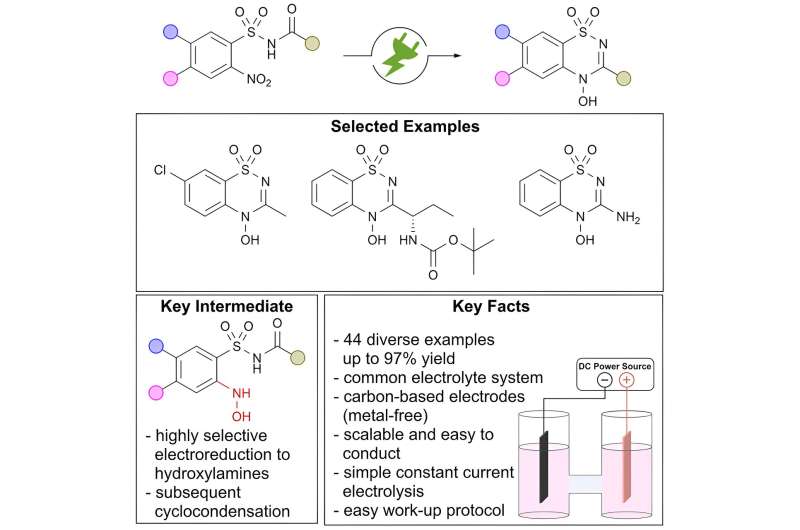
In a recent paper, a team of scientists from the Max Planck Institute of Chemical Energy Conversion (MPI CEC) shows how electrochemistry is driving the development of more sustainable synthetic routes for the modern organic chemist's toolkit by demonstrating that the electrochemical conversion of nitro groups, whose reduction normally requires large amounts of reductants or scarce metals, provides direct access to the previously understudied class of N-hydroxy heterocycles, an innovative structural motif for modern drug discovery.
Closing knowledge gaps—new findings on heterocycles
Nitrogen-containing heterocycles, in particular, benzo[e]-1,2,4-thiadiazine-1,1-dioxides, are a widely used structural motif in modern blockbuster pharmaceuticals, like Diazoxide. In contrast, little is known about heterocycles containing an exocyclic N-hydroxy modification. The high stability and the unique characteristics of the N-O bond make this motif highly interesting for modern pharmaceutical research. In addition, N-oxy heterocycles are often found as major drug metabolites.
Electrochemical synthesis by nitro reduction enables selective synthesis of this class of compounds, which is difficult to access by classical routes.
Benzo[e]-1,2,4-thiadiazine-1,1-dioxides represent a structural motif found in multiple APIs. However, synthetic approaches to access N-hydroxy derivatives containing a unique exocyclic N-O modification have not been reported so far.
Siegfried R. Waldvogel and his co-workers from MPI CEC and Johannes Gutenberg-Universität Mainz show in their recently published paper that electro-organic, a sustainable and powerful synthetic technique, enables highly selective and scalable access by cathodic reduction of easily accessible nitro arenes.
The team of scientists developed a highly selective and scalable electro-organic synthesis of novel benzo[e]-1,2,4-thiadiazine-1,1-dioxides, which represents an important structural motif of APIs modified with a unique N-hydroxy moiety.
Potential for use in metabolic research and pharmaceutical research
This study has enabled preparative access to novel substances bearing an N-hydroxy modification of a structural motif commonly found in drugs and expanded the compound library of benzo[e]-1,2,4-thiadiazine-1,1-dioxides. However, the biological properties of these compounds are still completely unknown. Moreover, their investigation could help to improve the efficiency of drugs, to understand metabolism, or to explore new pharmaceutical applications.
The paper is published in the journal Cell Reports Physical Science.
More information: Johannes Winter et al, Highly selective scalable electrosynthesis of 4-hydroxybenzo[e]-1,2,4-thiadiazine-1,1-dioxides, Cell Reports Physical Science (2024). DOI: 10.1016/j.xcrp.2024.101927
Journal information: Cell Reports Physical Science
Provided by Max Planck Society