This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Scientists explore the strategies of defects and nanostructure fabrication for promoting piezocatalytic activity
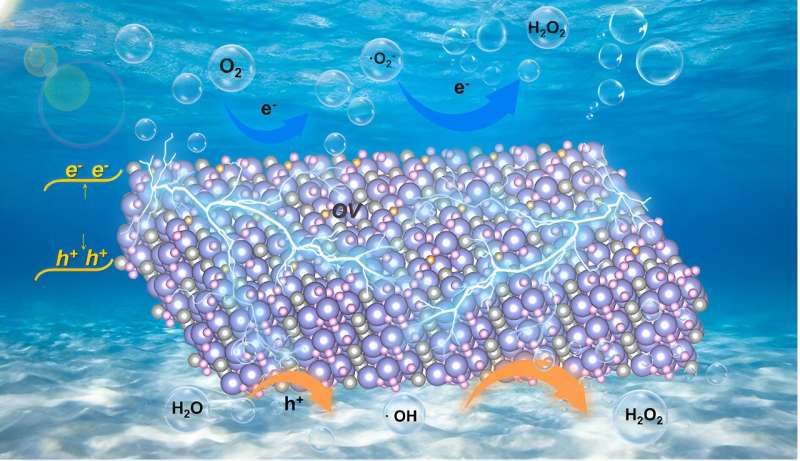
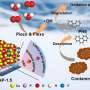
As an important chemical raw material, hydrogen peroxide (H2O2) is widely applied in various aspects of industry and life. The industrial anthraquinone method for H2O2 production has the serious flaws, such as high pollution and energy consumption. By using ubiquitous mechanical energy, piezocatalytic H2O2 evolution has been proven as a promising strategy, but its progress is hindered by unsatisfied energy conversion efficiency.
Bi4O5Br2 is regarded as a highly attractive photocatalytic material due to its unique sandwich structure, excellent chemical stability, good visible light capture ability and suitable band structure. Aspired by its non-centrosymmetric crystal structure, piezoelectric performance has begun to enter the vision of researchers recently.
However, its potential as an efficient piezocatalyst is far from being exploited, especially the impacts of defects on piezocatalysis and piezocatalytic H2O2 production over Bi4O5Br2 remains scanty. Thus, mechanical energy-driven piezocatalysis provides a promising method for H2O2 synthesis from pure water with great attraction.
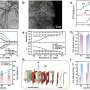
Recently, a research group led by Prof. Hongwei Huang from China University of Geosciences reported outstanding piezocatalytic H2O2 evolution performance that was achieved over ultrathin Bi4O5Br2 nanosheets with appropriate oxygen vacancies, and disclosed the mechanism that thin structure and oxygen vacancies collectively enhance the piezocatalytic activity.
The results were published in the Chinese Journal of Catalysis.
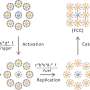
Ultrathin Bi4O5Br2 nanosheets with controllable oxygen vacancy concentrations are synthesized by a one-step solvothermal method by tuning the water to ethylene glycol ratio. Experiments and theoretical calculations have shown that Bi4O5Br2 with appropriate oxygen vacancies exhibits dramatic performance for piezocatalytic H2O2 production.
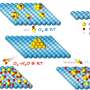
On the one hand, oxygen vacancies and thin structure largely increase the piezoelectric properties and piezoelectric potential of Bi4O5Br2, which improve the separation and transfer of piezoinduced charges. On the other hand, oxygen vacancies promote oxygen adsorption and activation on the surface of Bi4O5Br2, and lead to constantly decreased Gibbs free energy of the reaction pathway.
Therefore, the piezocatalytic H2O2 production performance of Bi4O5Br2 with appropriate oxygen vacancies is higher than that of other commonly used piezocatalysts.
More information: Hao Cai et al, Oxygen vacancies mediated ultrathin Bi4O5Br2 nanosheets for efficient piezocatalytic peroxide hydrogen generation in pure water, Chinese Journal of Catalysis (2024). DOI: 10.1016/S1872-2067(23)64591-7
Provided by Chinese Academy of Sciences