This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Light show in living cells: New method allows simultaneous fluorescent labeling of many proteins
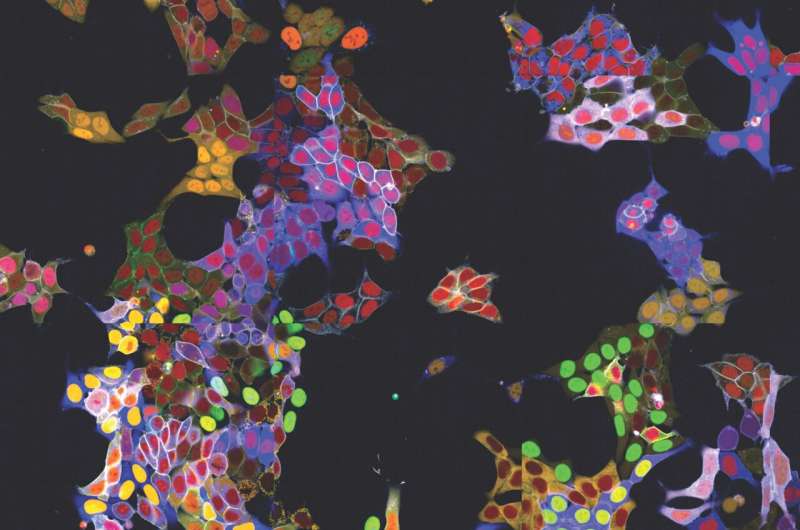
Observing proteins precisely within cells is extremely important for many branches of research but has been a significant technical challenge—especially in living cells, as the required fluorescent labeling had to be individually attached to each protein.
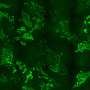
The research group led by Stefan Kubicek at CeMM has now overcome this hurdle: With a method called "vpCells," it is possible to label many proteins simultaneously, using five different fluorescent colors. This automated high-throughput approach, aided by AI-assisted image recognition, opens up entirely new applications in various disciplines, from fundamental cell biology to drug discovery. The study has been published in the journal Nature Cell Biology.
Without proteins, life as we know it would be inconceivable. They provide the structural framework for cells, act as enzymes to control metabolism, and enable cells to communicate with their environment as membrane receptors, transporters, or signaling molecules. All of these functions can only be fulfilled if the proteins are located in the right place within the cell. Often, even the properties of a protein change when it changes its location—control over its localization in the cell therefore also means control over its function.
To understand and explore the function of proteins, it is essential to precisely determine and track their location within the cell. Proteins often shuttle dynamically between different organelles and compartments of the cell. To visualize them under the microscope, they are often linked to a fluorescent, brightly shining protein component. However, this method has faced technical difficulties: Typically, the fluorescent component could only be attached to one protein at a time, and to label multiple proteins, cells usually had to be killed and fixed.
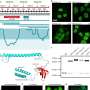
The new method presented by Stefan Kubicek's group, called "visual proteomics Cells" (abbreviated vpCells), allows proteins to be fluorescently labeled in a way that preserves their endogenous regulatory mechanisms. Instead of labeling one protein at a time, vpCells can fuse many proteins simultaneously with a fluorescent tag in a so-called multiplex approach.
A precursor of this method was already described by Kubicek's team in 2020 for studying metabolic enzymes. Now it has been expanded and improved in three ways:
Firstly, vpCells can label all theoretically possible proteins using the CRISPR/Cas9 gene-editing tool to genetically attach fluorescent proteins to the proteins under investigation. Kubicek's group has created a genome-wide "library" for this purpose, enabling the fluorescent marking and systematic functional exploration of all possible human proteins.
Secondly, vpCells use not only one fluorescent color but a total of five complementary colors. In each cell, two different proteins to be tracked are marked. Additionally, another color marking is used to better distinguish individual clones. And two further colors mark the cell nucleus and membrane to delineate individual cells better.
Thirdly, this color scheme enables not only to generate visually appealing images, but also to optically recognize and discriminate the different proteins . Normally, this requires complex DNA sequencing after imaging to determine which protein is labeled. The vpCells approach, on the other hand, enables training an AI-assisted image recognition system to recognize which protein is marked in which cell based solely on fluorescence microscopy images.
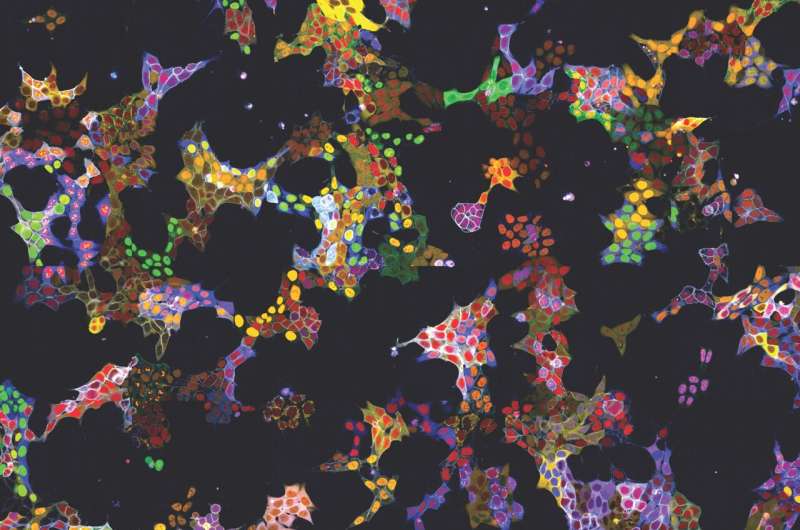
The method has already demonstrated its utility in two applications: On the one hand, more than 4,500 cell lines were generated as reporters for more than 1,100 proteins. These cell lines were used to train the AI models and to describe localization of the proteins in their basal state. All images of the individual labeled proteins are available on the publicly accessible web database vpCells.
On the other hand, the living reporter cells were used for a specific research question: Kubicek's team examined the effect of more than 1000 small-molecule substances on 61 proteins relevant to cancer cells. The researchers found that 44 of the tested substances altered the amount or localization of individual proteins within 6 hours. One of the substances turned out to be an inhibitor of protein transport from the cell nucleus, which has a similar effect to a clinically approved drug for multiple myeloma, a cancer of the blood-forming system.
"These results provide a first glimpse into the versatility of the vpCells method," says Kubicek. "We expect many more future applications, from fundamental cell biology to applied drug discovery."
More information: Pooled multicolor tagging for visualizing subcellular protein dynamics, Nature Cell Biology (2024). DOI: 10.1038/s41556-024-01407-w
Journal information: Nature Cell Biology
Provided by CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences