This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New catalyst accelerates release of hydrogen from ammonia
Germany can probably only meet its demand for climate-friendly hydrogen by imports, for example, from South America or Australia. For such long-distance transport, hydrogen can be converted into ammonia.
To facilitate the release of the hydrogen afterward, researchers from the Institute of Inorganic Chemistry at Kiel University (CAU) and their cooperation partners have developed a more active and cost-effective catalyst. The results were obtained as part of the hydrogen flagship project TransHyDE and have recently been published in Nature Communications.
The ability to store energy from wind or solar power plays a key role in the energy transition. "Storing energy in the form of chemical compounds such as hydrogen has many advantages. The energy density is high and the chemical industry also needs hydrogen for many processes," says Malte Behrens, Professor of Inorganic Chemistry at Kiel University. In addition, "green hydrogen" can be produced by electrolysis using electricity from renewable energy sources without producing CO2.
Infrastructure for ammonia already exists
But transporting hydrogen directly from regions where wind and solar power are cheap is not easy. An interesting alternative is the chemical conversion into ammonia. Ammonia itself contains a relatively high amount of hydrogen, and a well-developed infrastructure for its overseas transport already exists.
"Ammonia can be liquefied easily for transport, is already produced on a megaton scale and shipped and traded worldwide," says Dr. Shilong Chen, the leader of the Kiel subproject in the TransHyDE project "AmmoRef."
The two scientists from CAU's priority research area, KiNSIS (Kiel Nano, Surface and Interface Science), are collaborating with colleagues from Berlin, Essen, Karlsruhe, and Mülheim/Ruhr. Together, they are investigating how hydrogen can be catalytically released from ammonia after transport. Their newly developed catalyst significantly accelerates this reaction.
AmmoRef is one of ten TransHyDE projects. Scientists from a total of eight institutions are working on various sub-projects to improve hydrogen transport technologies. The results will be incorporated into the recommendations for the national hydrogen infrastructure.
Metal combination makes the catalyst highly active
"A catalyst accelerates a chemical reaction and is therefore directly responsible for the efficiency of chemical processes and energy conversion," Behrens explains. The faster the ammonia reforming process takes place, the lower the conversion losses caused by the chemical storage of hydrogen in ammonia.
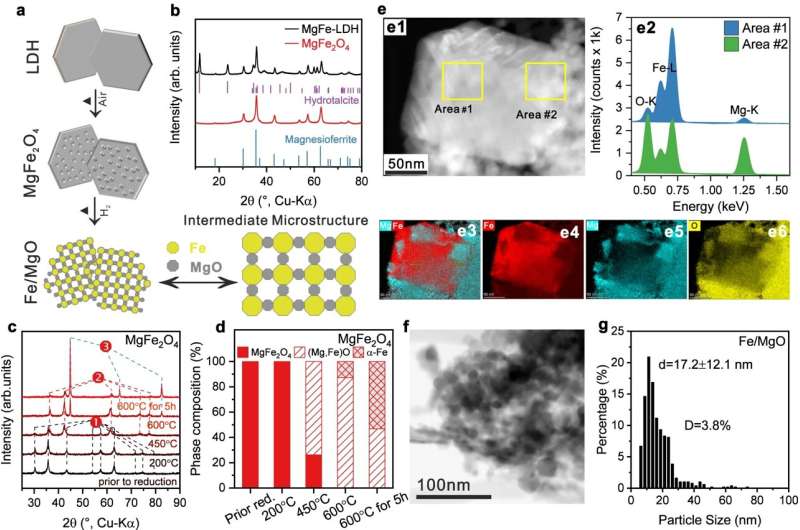
"Our catalyst has two special features," says Chen. "First, it is made of the relatively inexpensive base metals iron and cobalt. Secondly, we have developed a special synthesis process that allows a very high metal loading of this catalyst."
Up to 74% of the material consists of active metal nanoparticles, which are arranged between support particles in a way that cavities on the nano-scale are formed, looking like a porous metallic nano-sponge. "The combination of the two metals in an alloy is also crucial," Behrens explains. On their own, both metals are less catalytically active. The combination creates highly active bimetallic surfaces with properties that are otherwise only known from much more expensive precious metals.
"We will continue to investigate this catalyst in the AmmoRef consortium, in which industrial companies are also involved, and transfer it from basic research to application," says Behrens, announcing the next steps. To this end, the team in Kiel will now work on scaling up the synthesis.
More information: Shilong Chen et al, Highly loaded bimetallic iron-cobalt catalysts for hydrogen release from ammonia, Nature Communications (2024). DOI: 10.1038/s41467-023-44661-6
Provided by Kiel University