This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers observe highly excited 'roaming' energy pathway in chemical reactions
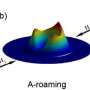
Scientists have observed so-called roaming chemical reactions, those that at certain points move away from the lowest minimum energy path of least resistance, in highly excited energy states for the first time.
Chemical reactions are supposed to occur along their minimum energy paths. In recent years, so-called roaming reactions that stray far from this path have begun to be observed, but only for chemical species in their ground state or, at most, their first excited state. However, researchers have now observed a roaming reaction even in highly excited energy states.
The researchers from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) described their findings in a paper appearing in Science.
Until recently, chemists had assumed that chemical reactions occur along what they call minimum energy paths—the reaction pathway that uses the lowest amount of energy between the starting stable configuration of a molecule and its final stable state.
At a certain point in any chemical reaction, there lies a transition state, at which the potential energy has a maximum value. This can be thought of as akin to a ball rolling up a hill and down again. But that transition state at the top of the hill still lies along the minimum energy path. Reactions arent supposed to stray away from this path of least resistance.
But in 2004, researchers exploring the breakdown of formaldehyde when being bombarded by photons (a chemical reaction called photodissociation) were shocked to discover that there are chemical reactions that can, in fact, stray well away from the minimum energy path.
This straying, or more properly roaming, happens when the expected cleavage of a chemical bond instead becomes frustrated: A component of a molecule begins to escape its parent molecule, but finds it does not have sufficient energy to do so. So instead, the component just orbits the remaining molecular fragment in a non-minimum-energy state.
It continues this orbit until it bumps into a reactive site (the physical location on a molecule where the reaction takes place and a new chemical bond is formed) of another molecule, returning to the minimum energy path.
Since then, these roaming reactions have been found to not just be occasional occurrences, but common ones.
"It turned out that roaming is a general aspect of chemical reactivity that had never been noticed before," said Fu Bina, one of the corresponding authors of the paper from DICP.
Further investigations have observed roaming reactions in both ground states—a molecule's lowest possible energy, and in their first excited states. When absorbing energy, an electron in a molecule jumps up to higher energy levels, called excited states. But roaming had only been observed in the first such excited states, not in any subsequent, higher excited states. Nor has roaming been observed to lead to the generation of electronically excited products of the chemical reaction.
However, the authors of the paper reported they had observed roaming in a highly excited state for the first time, in this case during the photodissociation of sulfur dioxide (SO2) molecules into sulfur and oxygen (a molecule of SO2 breaks down into an atom of sulfur, S, and one molecule of oxygen, O2, when bombarded by light).
Their results revealed two different possible pathways for dissociation. One proceeds along the expected minimum energy path to produce a vibrationally colder O2 molecule, and the other yields a vibrationally hotter O2 molecule in its electronically excited state.
"The latter reaction does this via a roaming pathway involving a sort of 'blob' of a single oxygen atom, what we call an 'intramolecular O- abstraction,' during a motion in which the molecule reorients itself," said Yuan Kaijun, another corresponding author of the paper from DICP.
Any time there is an increased probability of encountering frustrated bond cleavage, there is an increased likelihood of roaming reactions in highly excited states and production of electronically excited products. Such roaming dynamics may turn out to be the rule, the researchers believe, rather than the exception for molecular photodissociation through highly excited states.
The researchers were interested in SO2 in particular, given its importance in the Earth's atmosphere. Changes in the abundance of SO2 impact the radiation balance of the planet and thus the climate, and SO2 from volcanic eruptions is one of the two most important sources of aerosols in the stratosphere, and electronically excited products themselves react very differently in the atmosphere, in space, and in combustion.
Finally, photodissociation of SO2 could be of great importance for understanding the sources of molecular oxygen (O2) in the Earth's primitive atmosphere prior to the emergence of life.
As a result of their findings, the researchers argue that the roaming mechanism of molecular oxygen production should now be incorporated into photochemical modeling of the atmospheres of planets with rich volcanic outgassing of SO2.
More information: Zhenxing Li et al, Roaming in highly excited states: the central atom elimination of triatomic molecule decomposition, Science (2024). DOI: 10.1126/science.adn3357. www.science.org/doi/10.1126/science.adn3357
Journal information: Science
Provided by Chinese Academy of Sciences